Understanding the Foundation of Our Planet’s Crust
Every rock on Earth tells a story, and at the heart of that story lie minerals — the basic crystalline ingredients of our planet.
Among thousands of known minerals, only a small group forms the majority of Earth’s crust. These are the rock-forming minerals — the natural compounds that create the mountains, ocean floors, and continents we live on.

What Are Rock-Forming Minerals?
Rock-forming minerals are the most abundant minerals in the Earth’s crust and mantle.
They make up about 99% of the planet’s solid surface by weight.
These minerals crystallize directly from magma, form through metamorphic processes, or precipitate from water solutions — giving rise to igneous, metamorphic, and sedimentary rocks.
Most belong to a few key mineral families, particularly the silicates, which dominate the crust due to silicon and oxygen being the two most abundant elements on Earth.
Why They Matter
Rock-forming minerals are essential not only for geologists but also for engineers, environmental scientists, and anyone studying Earth’s systems.
They determine the composition, texture, color, and strength of rocks and influence processes like weathering, soil fertility, and even climate regulation through the carbon–silicate cycle.
Understanding these minerals allows us to read the geological past, predict resource distribution, and even study the formation of other planets.

The Major Classes of Rock-Forming Minerals
1. Silicate Minerals – The Dominant Group
Silicates account for over 90% of Earth’s crust.
Their structure is based on the silicon-oxygen tetrahedron (SiO₄), a versatile building block that can link together in various ways — creating a huge diversity of minerals.
| Silicate Sub-Group | Example Minerals | Typical Rock Types |
|---|---|---|
| Framework Silicates | Quartz, Feldspar | Granite, Sandstone |
| Sheet Silicates | Mica, Chlorite | Schist, Phyllite |
| Chain Silicates | Pyroxene, Amphibole | Basalt, Gabbro |
| Isolated Tetrahedra (Nesosilicates) | Olivine, Garnet | Peridotite, Basalt |
2. Non-Silicate Minerals
Although far less abundant, non-silicate minerals such as carbonates, oxides, sulfates, and halides play important roles in sedimentary and metamorphic environments.
Examples include calcite (CaCO₃), hematite (Fe₂O₃), gypsum (CaSO₄·2H₂O), and halite (NaCl).
These minerals often form through evaporation, precipitation, or alteration processes.
The Most Common Rock-Forming Minerals
Let’s explore the key minerals that literally build our planet:

Quartz (SiO₂)
- Type: Framework silicate
- Color: Colorless, white, or tinted by impurities (amethyst, smoky, rose)
- Hardness: 7 (Mohs scale)
- Occurrence: Igneous, metamorphic, and sedimentary rocks
Quartz is chemically resistant and physically hard — one of the most durable minerals on Earth.
It forms the bulk of sand grains, granites, and metamorphic quartzites.
Because of its stability, quartz remains even after other minerals weather away.
? See more: Quartz Mineral
Feldspar Group
- Type: Framework silicate
- Major Members: Orthoclase (K-feldspar), Plagioclase (Na-Ca feldspar)
- Occurrence: Igneous and metamorphic rocks
- Crustal Abundance: ~60% of continental crust
Feldspars are the most abundant minerals in the crust. They crystallize from magma and later weather to form clays.
Orthoclase gives granites their pinkish hue, while plagioclase appears in basalts and gabbros.
? See more: Feldspar Minerals
Mica Group
- Type: Sheet silicate
- Major Members: Muscovite (light), Biotite (dark)
- Properties: Perfect cleavage into thin elastic sheets
- Occurrence: Common in granites, schists, and pegmatites
Micas are prized for their flexibility and shiny appearance.
They form in both igneous and metamorphic settings and help geologists understand pressure–temperature histories in rocks.
? See more: Mica Minerals
Amphibole Group
- Type: Double chain silicate
- Example: Hornblende
- Occurrence: Intermediate to felsic igneous rocks and metamorphic schists
Amphiboles are complex silicates containing iron, magnesium, calcium, and sometimes sodium.
They typically form under moderate to high metamorphic conditions and help determine the metamorphic grade of rocks.
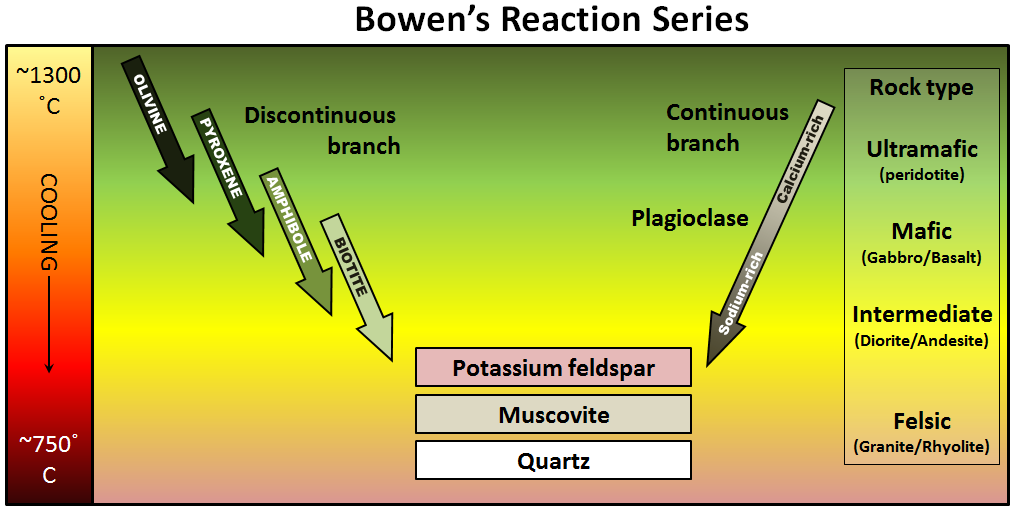
Pyroxene Group
- Type: Single chain silicate
- Examples: Augite, Diopside
- Occurrence: Basalt, gabbro, peridotite
Pyroxenes crystallize in high-temperature conditions and dominate the upper mantle.
They are key indicators in studying mantle xenoliths brought up by volcanic eruptions.
Olivine (Mg,Fe)₂SiO₄
- Type: Isolated tetrahedra silicate (nesosilicate)
- Color: Olive green to yellowish
- Occurrence: Mantle rocks (peridotite), basaltic lavas
Olivine is one of the first minerals to crystallize from magma and one of the most common in the upper mantle.
It plays a crucial role in understanding mantle convection and magmatic differentiation.
? See more: Olivine Mineral
Calcite (CaCO₃)
- Type: Carbonate
- Occurrence: Sedimentary rocks (limestone, chalk), metamorphic marble
Calcite is not a silicate but remains one of the most abundant minerals near the Earth’s surface.
It reacts easily with acids and forms many karst landscapes through dissolution.
? Related: Limestone Rock

Mineral Associations and Rock Types
How Rock-Forming Minerals Shape Rocks
The combination and proportion of these minerals define rock types:
| Rock Type | Dominant Minerals |
|---|---|
| Granite | Quartz, K-Feldspar, Mica |
| Basalt | Pyroxene, Plagioclase, Olivine |
| Gneiss | Feldspar, Quartz, Biotite |
| Marble | Calcite |
| Sandstone | Quartz, Feldspar |
| Peridotite | Olivine, Pyroxene |
Each mineral crystallizes under specific temperature and pressure conditions, described by Bowen’s Reaction Series — a key principle in petrology that explains how igneous rocks form from cooling magma.
Rock-Forming Minerals and the Carbon Cycle
Certain rock-forming minerals, especially silicates, help regulate Earth’s long-term climate.
When silicates weather, they react with atmospheric CO₂, removing it from the air and storing carbon in carbonate rocks.
This process, though slow, acts as a global thermostat, balancing greenhouse gases over millions of years. for understanding the processes that occur during rock formation and the conditions under which rocks have formed. It is an essential aspect of petrology, which is the branch of geology that deals with the study of rocks and their mineralogical composition.

Formation and Alteration of Rock-Forming Minerals
Formation of Rock-Forming Minerals: Rock-forming minerals can form through various processes depending on the type of rock and the geological conditions. Some common processes of mineral formation include:
- Crystallization from Magma: Many rock-forming minerals form by crystallization from a magma (molten rock) during the process of cooling and solidification. As the magma cools, minerals with different melting points crystallize and solidify at different temperatures, leading to the formation of distinct mineral assemblages. For example, minerals like quartz, feldspars, and mica can form from the cooling of a magma to form igneous rocks such as granite.
- Precipitation from Solution: Minerals can also form through the precipitation of dissolved ions from a solution. This process can occur in various environments, such as lakes, seas, hot springs, and hydrothermal vents. As the concentration of dissolved ions in the solution changes due to changes in temperature, pressure, or chemical composition, minerals can precipitate and accumulate to form sedimentary rocks such as limestone, halite, and gypsum.
- Metamorphism: Metamorphism is the process by which existing rocks undergo changes in mineralogical composition and/or texture due to high temperature, pressure, and/or chemical alteration. During metamorphism, existing minerals in a rock can recrystallize, react with other minerals, and/or undergo changes in their mineralogy. For example, the mineral mica in a shale can transform into the mineral chlorite during metamorphism, forming a new rock called a chlorite schist.
Alteration of Rock-Forming Minerals: Rock-forming minerals can also undergo alteration or weathering due to various geological processes, such as chemical reactions, dissolution, hydrolysis, oxidation, and reduction. Some common types of mineral alteration include:
- Weathering: Weathering is the process by which rocks and minerals at or near the Earth’s surface break down and undergo chemical and physical changes due to exposure to air, water, and other environmental factors. This can result in the alteration of rock-forming minerals, such as the breakdown of feldspars into clay minerals during chemical weathering.
- Diagenesis: Diagenesis is the process by which sedimentary rocks undergo changes in mineralogical composition and texture due to compaction, cementation, and other chemical reactions. This can result in the alteration of minerals in sedimentary rocks, such as the formation of cementing minerals (e.g., silica, calcium carbonate) that bind sediment grains together.
- Hydrothermal Alteration: Hydrothermal alteration occurs when rocks and minerals come into contact with hot fluids, such as groundwater or magma, which can cause chemical reactions and alteration of minerals. This process can lead to the formation of new minerals and alteration of existing minerals, resulting in changes in the mineralogy and texture of the rocks. For example, hydrothermal alteration can form minerals like quartz, sericite, and chlorite in rocks such as hydrothermal veins and altered volcanic rocks.
The formation and alteration of rock-forming minerals are important processes in the geologic cycle and play a crucial role in the formation, composition, and characteristics of rocks. They also have significant implications for various geologic processes, such as mineral resource formation, soil formation, and environmental processes, and are studied in detail in the field of mineralogy and petrology within geology.
Economic and Industrial Uses
| Mineral | Common Uses |
|---|---|
| Quartz | Glass, electronics, solar cells |
| Feldspar | Ceramics, enamel, glassmaking |
| Mica | Electrical insulators, cosmetics |
| Olivine | Foundry sand, refractory material |
| Calcite | Cement, lime, acid neutralization |
These minerals, though ordinary in geology, are extraordinary in utility — linking deep-Earth processes with everyday human life.
Summary Table: Key Rock-Forming Minerals
| Mineral | Chemical Formula | Mineral Group | Rock Type | Hardness (Mohs) |
|---|---|---|---|---|
| Quartz | SiO₂ | Silicate | Igneous, Metamorphic, Sedimentary | 7 |
| Feldspar | KAlSi₃O₈–NaAlSi₃O₈–CaAl₂Si₂O₈ | Silicate | Igneous, Metamorphic | 6 |
| Mica | KAl₂(AlSi₃O₁₀)(F,OH)₂ | Silicate | Igneous, Metamorphic | 2–3 |
| Amphibole | Complex (Ca,Na)(Mg,Fe,Al)₅(Si,Al)₈O₂₂(OH)₂ | Silicate | Igneous, Metamorphic | 5–6 |
| Pyroxene | (Mg,Fe,Ca)SiO₃ | Silicate | Igneous | 5–6 |
| Olivine | (Mg,Fe)₂SiO₄ | Silicate | Igneous | 6.5–7 |
| Calcite | CaCO₃ | Carbonate | Sedimentary, Metamorphic | 3 |
References
- Geology.com – Rock-Forming Minerals: The Building Blocks of the Earth’s Crust
- LibreTexts – Fundamentals of Geology, Chapter 2: Rock-Forming Minerals
- OpenGeology.org – Earth Materials and Mineral Composition of Rocks
- Encyclopaedia Britannica – Mineral (Geology)
- Mindat.org – Rock-Forming Minerals Database
- GIA (Gemological Institute of America) – Introduction to Common Minerals
- Klein, C. & Dutrow, B. (2012). Manual of Mineral Science. Wiley-Blackwell.
- Nesse, W. D. (2017). Introduction to Mineralogy (3rd ed.). Oxford University Press.
- Deer, W. A., Howie, R. A., & Zussman, J. (2013). An Introduction to the Rock-Forming Minerals (3rd ed.). The Mineralogical Society of Great Britain and Ireland.
- Hurlbut, C. S. & Klein, C. (1985). Manual of Mineralogy (20th ed.). John Wiley & Sons.