Feldspar is the name of a large organization of rock-forming silicate minerals that make up over 50% of Earth’s crust. They are discovered in igneous, metamorphic, and sedimentary rocks in all components of the sector. Feldspar minerals have very comparable structures, chemical compositions, and bodily properties. Common feldspars consist of orthoclase (KAlSi3O8), albite (NaAlSi3O8), and anorthite (CaAl2Si2O8).
Compositions of Feldspar Group Minerals
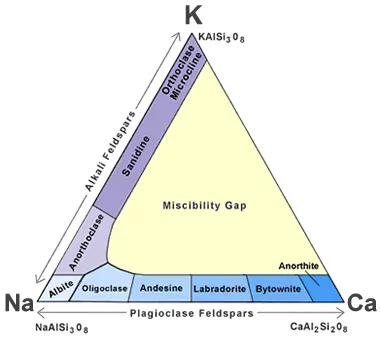
This group of minerals includes tectosilicates. Compositions of foremost elements in commonplace feldspars may be expressed in terms of 3 endmembers: potassium feldspar (K-spar) endmember KAlSi3O8, albite endmember NaAlSi3O8, anorthite endmember CaAl2Si2O8. Solid answers between K-feldspar and albite are referred to as “alkali feldspar”. Solid solutions among albite and anorthite are called “plagioclase”,or greater nicely “plagioclase feldspar”. Only constrained solid answer happens between K-feldspar and anorthite, and inside the two different stable answers, immiscibility occurs at temperatures commonplace in the crust of the Earth. Albite is taken into consideration both a plagioclase and alkali feldspar.
Physical Properties of Feldspar Minerals
| Chemical Classification | Silicate |
| Color | Usually white, pink, gray or brown. Also colorless, yellow, orange, red, black, blue, green. |
| Streak | White |
| Luster | Vitreous. Pearly on some cleavage faces. |
| Diaphaneity | Usually translucent to opaque. Rarely transparent. |
| Cleavage | Perfect in two directions. Cleavage planes usually intersect at or close to a 90 degree angle. |
| Mohs Hardness | 6 to 6.5 |
| Specific Gravity | 2.5 to 2.8 |
| Diagnostic Properties | Perfect cleavage, with cleavage faces usually intersecting at or close to 90 degrees. Consistent hardness, specific gravity and pearly luster on cleavage faces. |
| Chemical Composition | A generalized chemical composition of X(Al,Si)4O8, where X is usually potassium, sodium, or calcium, but rarely can be barium, rubidium, or strontium. |
| Crystal System | Triclinic, monoclinic |
| Uses | Crushed and powdered feldspar are important raw materials for the manufacture of plate glass, container glass, ceramic products, paints, plastics and many other products. Varieties of orthoclase, labradorite, oligoclase, microcline and other feldspar minerals have been cut and used as faceted and cabochon gems. |
Alkali Feldspar Minerals
The alkali feldspars are as follows:
- Orthoclase (Monoclinic)[10] KAlSi3O8,
- Sanidine (Monoclinic)[11] (K,Na)AlSi3O8,
- Microcline (Triclinic)[12] KAlSi3O8,
- Anorthoclase (Triclinic) (Na,K)AlSi3O8.
Sanidine is stable at the highest temperatures, and microcline at the lowest. Perthite is a typical texture in alkali feldspar, due to exsolution of contrasting alkali feldspar compositions during cooling of an intermediate composition. The perthitic textures in the alkali feldspars of many granites can be seen with the naked eye.Microperthitic textures in crystals are visible using a light microscope, whereas cryptoperthitic textures can be seen only with an electron microscope.
Many Types of Feldspar
| Mineral | Composition |
| Albite | NaAlSi3O8 |
| Amazonite | KAlSi3O8 |
| Andesine | (Na,Ca)(Al,Si)4O8 |
| Anorthite | CaAl2Si2O8 |
| Anorthoclase | (Na,K)AlSi3O8 |
| Banalsite | Na2BaAl4Si4O16 |
| Buddingtonite | (NH4)AlSi3O8 |
| Bytownite | (Ca,Na)(Al,Si)4O8 |
| Celsian | BaAl2Si2O8 |
| Dmisteinbergite | CaAl2Si2O8 |
| Filatovite | K(Al,Zn)2(As,Si)2O8 |
| Hexacelsian | BaAl2Si2O8 |
| Hyalophane | (K,Ba)(Al,Si)4O8 |
| Kokchetavite | KAlSi3O8 |
| Kumdykolite | NaAlSi3O8 |
| Labradorite | (Ca,Na)(Al,Si)4O8 |
| Microcline | KAlSi3O8 |
| Oligoclase | (Na,Ca)(Al,Si)4O8 |
| Orthoclase | KAlSi3O8 |
| Paracelsian | BaAl2Si2O8 |
| Reedmergnerite | NaBSi3O8 |
| Rubicline | (Rb,K)AlSi3O8 |
| Sanidine | KAlSi3O8 |
| Slawsonite | SrAl2Si2O8 |
| Stronalsite | Na2SrAl4Si4O16 |
| Svyatoslavite | CaAl2Si2O8 |
Barium feldspars
Barium feldspars are also considered alkali feldspars. Barium feldspars form as the result of the substitution of barium for potassium in the mineral structure. The barium feldspars are monoclinic and include the following:
- Celsian BaAl2Si2O8,
- Hyalophane (K,Ba)(Al,Si)4O8.
Plagioclase feldspars
| Plagioclase Mineral Name | Percent NaAlSi3O8 | Percent CaAl2Si2O8 |
| Albite | 100-90% albite | 0-10% anorthite |
| Oligoclase | 90-70% albite | 10-30% anorthite |
| Andesine | 70-50% albite | 30-50% anorthite |
| Labradorite | 50-30% albite | 50-70% anorthite |
| Bytownite | 30-10% albite | 70-90% anorthite |
| Anorthite | 10-0% albite | 90-100% anorthite |
The plagioclase feldspars are triclinic. The plagioclase series follows (with percent anorthite in parentheses):
Albite (0 to 10) NaAlSi3O8,
Oligoclase (10 to 30) (Na,Ca)(Al,Si)AlSi2O8,
Andesine (30 to 50) NaAlSi3O8—CaAl2Si2O8,
Labradorite (50 to 70) (Ca,Na)Al(Al,Si)Si2O8,
Bytownite (70 to 90) (NaSi,CaAl)AlSi2O8,
Anorthite (90 to 100) CaAl2Si2O8.
Production and Uses of Feldspar Minerals
About 20 million tonnes of feldspar have been produced in 2010, primarily by three countries: Italy (four.7 Mt), Turkey (4.Five Mt), and China (2 Mt)
Feldspar is a common uncooked fabric utilized in glassmaking, ceramics, and to a point as a filler and extender in paint, plastics, and rubber. In glassmaking, alumina from feldspar improves product hardness, sturdiness, and resistance to chemical corrosion. In ceramics, the alkalis in feldspar (calcium oxide, potassium oxide, and sodium oxide) act as a flux, decreasing the melting temperature of a combination. Fluxes melt at an early stage in the firing method, forming a glassy matrix that bonds the opposite additives of the gadget collectively. In the US, approximately sixty six% of feldspar is consumed in glassmaking, including glass containers and glass fiber. Ceramics (inclusive of electric insulators, sanitaryware, pottery, tableware, and tile) and different uses, which includes fillers, accounted for the remainder.