Geology, Uses & the Global Supply Crisis Shaping Our Technological Future
Rare earth minerals are one of the strangest contradictions in modern geology: visually unimpressive, chemically tricky, and geologically scattered—yet absolutely essential to almost every piece of technology we rely on today. Smartphones, electric vehicles, wind turbines, satellites, medical imaging systems, laser equipment, and even advanced military technologies depend on them. Without rare earth elements, the modern digital world simply could not function.
And the irony? These elements are not truly “rare” in the Earth’s crust. Many are actually more abundant than precious metals like gold. The problem is that they almost never occur in concentrated, mineable deposits, and separating them from surrounding minerals requires extremely complex chemistry. As a result, global production has become dangerously centralized, pushing the world into a quietly growing supply crisis.
This article takes you through how rare earth minerals form, why industries are desperate for them, and how the geopolitical imbalance surrounding their supply is shaping the future of global technology.
1. What Exactly Are Rare Earth Elements—and Why Aren’t They Really Rare?

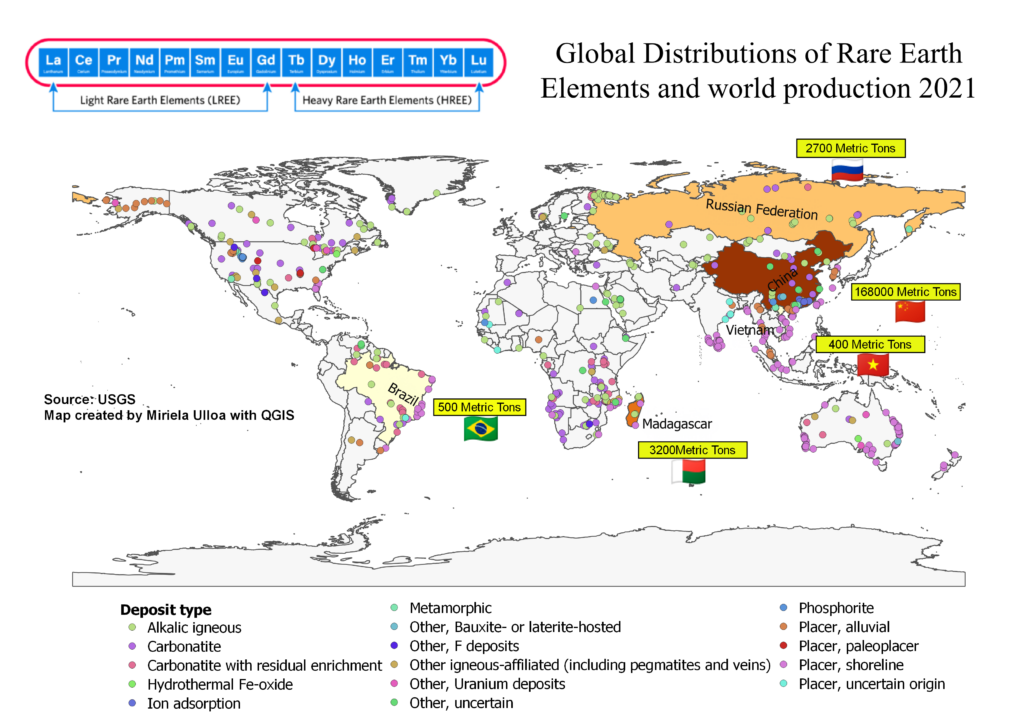
The rare earth group consists of 17 elements: the 15 lanthanides, plus yttrium and scandium. Chemically, they behave so similarly that they often occur together in nature, substituting for one another within crystal structures. This chemical similarity is a blessing for advanced technology—but a curse for mining companies trying to separate them.
REEs typically accumulate in specific geological environments:
- Alkali igneous complexes
- Carbonatite intrusions
- Pegmatite systems
- Hydrothermal alteration halos
- Ion-adsorption clays in tropical regions
Their abundance is not the issue; economic concentration is. Most crustal rocks contain trace amounts of REEs, but only a few geological processes enrich them enough to form an ore deposit. And even in those deposits, extracting and refining them is a multi-stage, chemically intensive process that many countries are reluctant to undertake.
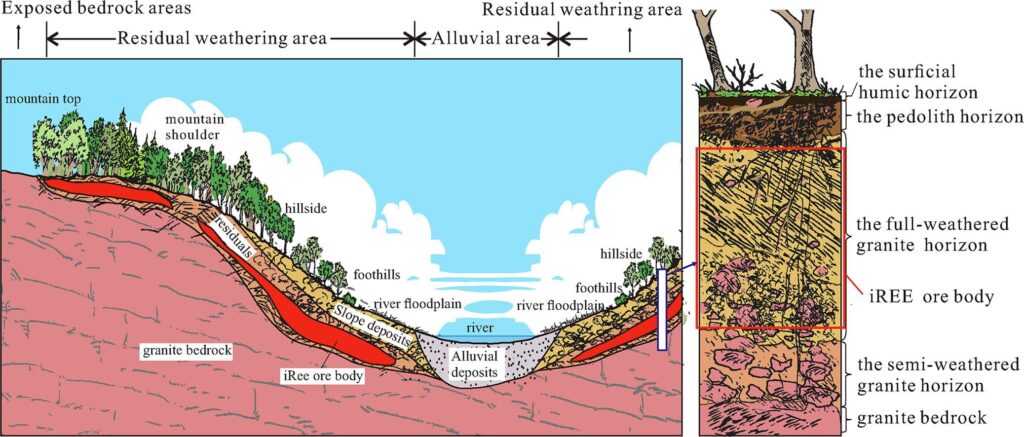
2. How Rare Earth Minerals Form: From Magma to Weathered Clay

a) Magmatic origins
Some of the world’s most important REE deposits form in unusual igneous settings—especially karbonatites and alkali complexes. These magmas contain high concentrations of volatile components (CO₂, fluorine, chlorine) that keep rare earth elements dissolved until the very last stages of crystallization.
The most economically important REE-bearing minerals include:
- Bastnäsite – a fluorocarbonate rich in cerium, lanthanum, and neodymium
- Monazite – a phosphate mineral containing light REEs and often thorium
- Xenotime – a yttrium-rich phosphate
These minerals crystallize in small but valuable pockets within the igneous body.
b) Pegmatites
Pegmatites form from the final, highly enriched melt of a crystallizing magma chamber. This melt is loaded with water, volatiles, and incompatible elements—perfect conditions for REE-rich minerals to grow. Some pegmatites hold exceptionally high concentrations of neodymium, dysprosium, and other strategic metals.
c) Hydrothermal alteration systems
Circulating hot fluids can dissolve REEs and redeposit them in mineralized halos around igneous intrusions. These zones may contain xenotime, monazite, or complex REE-bearing silicates produced through fluid-rock reactions.
d) Ion-adsorption clays
In tropical climates, prolonged weathering breaks down primary REE minerals and releases the elements into soil systems. Clay minerals trap REEs on their surfaces through ion exchange. These deposits, especially in southern China, produce a large portion of the world’s heavy REEs and are much easier to process chemically than hard-rock ores.
3. Why Rare Earth Minerals Are Critical to Modern Technology
It’s impossible to understand the global dependence on rare earth minerals without looking at how deeply embedded they are in every major technology sector.
a) Electric vehicles
High-performance permanent magnets rely on neodymium, praseodymium, and dysprosium. These magnets are incredibly strong for their size, which is why EV motors can be compact yet powerful. Without REE magnets, electric vehicles would be heavier, slower, and less efficient.
b) Renewable energy systems
Wind turbines use massive permanent magnets containing REEs. These magnets allow turbines to generate strong electrical output without complex gear systems. Large-scale green energy expansion depends directly on REE supply stability.
c) Smartphones and consumer electronics
Inside every phone and laptop:
- Speakers and vibration motors use REE magnets
- Display phosphors rely on europium and terbium
- Optical fibers use erbium for signal amplification
- Microchips contain trace REE alloys
Modern electronics are unimaginable without them.
d) Aerospace and defense
Rare earth elements are embedded in the defense infrastructure of nearly every advanced nation:
- Laser-targeting systems
- Missile guidance components
- Satellite communications
- Jet engine alloys
- Radar and sonar systems
These technologies require specific REE-based alloys and phosphors that have no substitutes.
e) Medical applications
Gadolinium is essential for MRI contrast agents. Terbium and europium produce high-quality illumination in imaging screens. Some lanthanides are even being tested in cancer treatments.
Rare earths are not luxury minerals—they are the backbone of civilization’s most advanced tools.

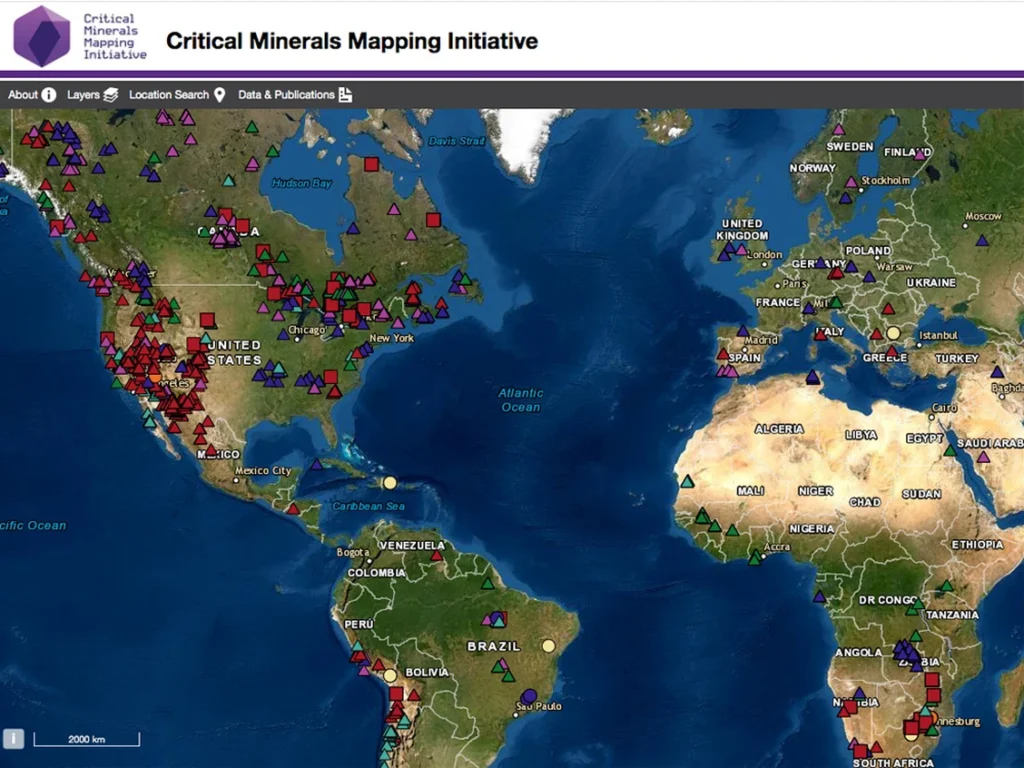
4. Global Production: Who Controls the World’s Supply?
Although REEs are widely distributed, only a few locations have deposits rich enough to mine. Over time, this geological fact, combined with massive industrial investment, has created a heavily unbalanced global supply network.
Current global reality:
- China produces 60–70% of the world’s mined REEs
- China refines 85% of global rare earth oxides
- China manufactures over 90% of high-strength REE magnets
Other countries, even when they mine REEs, still ship the ore to China for chemical processing. This gives China an unparalleled level of control over the global supply chain.
Major alternative deposits exist in:
- Mountain Pass, USA
- Mt. Weld, Australia
- Norra Kärr, Sweden
- Kvanefjeld, Greenland
- Ngualla, Tanzania
But refining capacity—not mining—is the true bottleneck.
5. Why the World Faces a Rare Earth Supply Crisis
Several deep-rooted issues make the REE supply chain fragile:
a) Refining is environmentally difficult and extremely expensive
Chemically separating 17 nearly identical elements requires multiple rounds of solvent extraction, strong acids, high energy consumption, and careful handling of radioactive by-products like thorium. Many countries avoid investing in this infrastructure for environmental and political reasons.
b) China undercut global competitors for decades
For nearly 20 years, China sold REEs at very low prices, pushing almost every competitor out of the market. Refining facilities closed across the U.S., Australia, and Europe. By the time demand skyrocketed, China already controlled the entire value chain.
c) The real power lies in refining—not mining
A country can discover a large REE deposit, but without refining capacity, it remains dependent on external processors. This is why global diversification has been so slow.
d) Geopolitical leverage
China has previously restricted REE exports during political disputes. This single move demonstrated how easily the supply chain can be weaponized, prompting the U.S., Japan, and the EU to classify REEs as “critical minerals.”

6. The Future: New Sources, New Technologies, and a Slow Escape from Dependence
To reduce strategic vulnerability, countries are developing new mines and rebuilding refining capacity.
Promising developments:
- The Mountain Pass mine in the U.S. is ramping up production again.
- Lynas in Australia is now the world’s largest non-Chinese REE producer.
- New carbonatite deposits in Africa show enormous potential.
- Greenland and northern Europe are exploring large-scale projects.
Recycling is emerging as a long-term solution, although it currently supplies only a small portion of global demand. In the distant future, deep-sea nodules may become viable, but environmental concerns remain significant.
Researchers are also exploring:
- REE-free motor designs
- More efficient magnet technologies
- Environmentally cleaner extraction methods
But none of these are ready to replace traditional production in the short term.
7. Conclusion: Our Technological World Runs on These Unassuming Minerals
Rare earth minerals may look ordinary, but they hold extraordinary power. Nearly every advanced system humans have built—communications networks, navigation satellites, medical imaging devices, renewable energy infrastructure, electric transport, aerospace engineering—depends on them.
And because refining is concentrated in one region of the world, the global economy is exposed to a single point of failure.
Nations are racing to secure their own supplies, develop new extraction technologies, and rebuild lost industrial capacity. But for now, rare earth elements remain one of the most strategically important—and most vulnerable—resources on the planet.