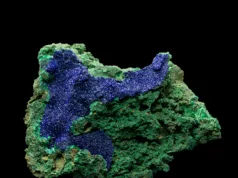
Carbonates Minerals
Carbonate minerals are a fascinating and widely occurring family built around the carbonate anion (CO₃²⁻), and they hold a special place in both Earth’s geology and human industry. From the marble and limestone that form majestic mountain ranges to the tiny shells of marine organisms that accumulate into thick sedimentary sequences, minerals like Calcite (CaCO₃), Dolomite (CaMg(CO₃)₂) and Magnesite (MgCO₃) tell stories of ancient seas, shifting chemical conditions and evolving life. These minerals form in diverse settings: shallow warm oceans, evaporitic lagoons, caves, hydrothermal veins and even in weathering zones. Their crystal structure, chemistry and textures record fluid chemistry, temperature, pressure and biological activity. In the field of engineering and construction, carbonate minerals matter greatly — crushed limestone is foundational for concrete and road aggregate, while dolomite serves as a refractory material and magnesite as a high-temperature brick. For geologists and site professionals, recognising carbonate minerals means understanding facies, diagenetic processes, rock stability, dissolution/precipitation behaviour and how ‘soft’ carbonate rock might behave differently from silicate rock in excavation or foundation work. In this category you’ll dive into how carbonate minerals form, how their chemistry adapts (e.g., substitution of Ca by Mg or Fe), how to spot them in hand sample or core, and why they remain vital both for reading Earth’s history and for practical engineering in the present day.
Chrysanthemum Stone
Chrysanthemum stone is a unique and captivating mineral with a rich history, valued for its striking appearance and intricate patterns. This stone is not...
Cobaltocalcite
Cobaltocalcite is a captivating and visually distinctive mineral that belongs to the calcite group of minerals. It is renowned for its remarkable pink to...
Smithsonite
Smithsonite is a mineral that belongs to the carbonate group. It is named after James Smithson, an English scientist and mineralogist who was the...
Cerussite
Cerussite is a mineral that belongs to the carbonate group. It is composed of lead carbonate (PbCO3) and has a relatively high lead content....
Aragonite
Aragonite is a carbonate mineral and its formula is calcium carbonate. It has the same formula as Calcite and Vaterite, but has a different...
Rhodochrosite
Rhodochrosite is a carbonate mineral with formula: MnCO3. It has a classic rose-pink color, but specimens can also be brown or gray. It forms dogtooth or rhombohedral crystals like calcite, but it may also occur in stalactitic, granular, nodular, botryoidal, and massive habits. Rhodochrosite is found in hydrothermal ore veins with sphalerite, galena, fluorite, and manganese oxides.
Malachite
Possibly the earliest ore of copper, malachite is believed to have been mined in the Sinai and eastern deserts of ancient Egypt from as early as 3000 BCE. Single crystals are uncommon; when found, they are short to long prisms.
Dolostone (Dolomite)
An important rock-forming mineral, dolomite is named after the French mineralogist Déodat Gratet de Dolomieu. It is a colorless to white, pale brown, grayish, reddish, or pink mineral. Its crystals are commonly rhombohedral or tabular, often have curved faces, and sometimes cluster in saddle-shaped aggregates.
Calcite
Calcite is a rock-forming mineral with a chemical formula of CaCO3. It is extremely common and found throughout the world in sedimentary, metamorphic, and igneous rocks. Some geologists consider it to be a "ubiquitous mineral" - one that is found everywhere.