Olivine: Green Mineral Coming from Earth’s Depths

Olivine is one of minerals that most people see without noticing in their lives but don’t know name of. Sometimes appears as small green grains inside volcanic rocks close to black. Sometimes shines inside beach sand. Sometimes also appears with name “peridot” in jewelry showcases.
But what really makes Olivine interesting is not its color or shine; it’s where it comes from.
This mineral is not ordinary stone formed on earth’s surface. Olivine is born in depths of our planet, in mantle. So when you take it in hand, you actually touch piece coming from inside Earth.
What Is Olivine?

Olivine is silicate mineral rich in magnesium and iron. Has quite fundamental place in mineralogy because is one of main components of Earth’s upper mantle. Meaning big part of our planet is rich in terms of olivine.
Color scale generally revolves around tones of green. Can vary from light yellowish green to dark olive green. These color differences are directly related to amount of iron mineral contains. As iron increases color darkens.
Crystal form is most of time not clear. Olivine is generally found in grain form. However developed crystals can also be seen under suitable conditions. These crystals have glass shine and offer lively appearance under light.
Name: Olivine derives its name from the usual olive-green color of the mineral, and is the term usually given to the species when speaking of it as a rock-forming mineral. Peridot is an old name for the species.
Alteration: Very readily altered to serpentine and less commonly to iddingsite. Magnesite and iron oxides may form at the same time as a result of the alteration.
Diagnostic Features: Distinguished usually by its glassy luster, conchoidal fracture, green color, and granular nature.
Composition: Silicate of magnesium and ferrous iron, (Mg,Fe)2Si0 4 . A complete isomorphous series exists, grading from forsterite, Mg2Si04, to fayalite, Fe2Si04. The more common olivines are richer in magnesium than in iron
Crystallography: Orthorhombic; dipyramidal. Crystals usually a combination of prism, macro- and brachypinacoids and domes, pyramid and base. Often flattened parallel to either the macro- or brachypinacoid. Usually in imbedded grains or in granular masses.
Where and How Does Olivine Form?

Story of Olivine starts not on Earth’s surface but in depths. This mineral crystallizes in mantle under high temperature and pressure conditions. Meaning normally is not possible for it to form on earth’s surface.
However volcanic activities carry this mineral to surface. While magma rises, brings olivine crystals inside it along. Green olivine grains we see in volcanic rocks are actually pieces broken off from mantle.
That’s why olivine is frequently found together with igneous rocks like:
- Basalt
- Peridotite
- Gabbro
Especially peridotite takes its name directly from olivine and is one of most characteristic rocks of mantle.
Place of Olivine in Bowen Reaction Series
Olivine has special position among igneous minerals. Because is one of first minerals forming at high temperatures.
According to Bowen Reaction Series, when magma starts cooling, first mineral to crystallize is olivine. This tells us this: Olivine is mineral of high temperature environments. Doesn’t love cold and calm conditions.
This feature also explains why olivine is unstable in surface conditions. Meaning this mineral doesn’t like staying on Earth’s surface for long time; undergoes chemical changes over time.
Olivine Composition
Olivine is the name given to a set of silicate minerals which have a generalized chemical composition of A2SiO4. In that generalized composition, “A” is generally Mg or Fe, however in unusual situations can be Ca, Mn, or Ni.
The chemical composition of most olivine falls somewhere between pure forsterite (Mg2SiO4) and pure fayalite (Fe2SiO4). In that series, Mg and Fe can alternative freely for each other in the mineral’s atomic structure – in any ratio. This form of non-stop compositional variation is called a “strong solution” and is represented in a chemical components as (Mg,Fe)2SiO4.
| Mineral | Chemical Composition |
| Forsterite | Mg2SiO4 |
| Fayalite | Fe2SiO4 |
| Monticellite | CaMgSiO4 |
| Kirschsteinite | CaFeSiO4 |
| Tephroite | Mn2SiO4 |
Why Does Olivine Deteriorate Rapidly on Surface?
Olivine is very stable in mantle. But when comes to surface things change.
When contacts with rain, oxygen and water, olivine slowly starts transforming into other minerals. In this process minerals like serpentine can form. That’s why rocks containing olivine generally look “fresh”; they can lose their green colors over time.
This feature makes olivine very valuable for geologists. Because fresh olivine crystals found on surface can be sign of volcanic activities that occurred in recent past.
Olivine and Peridot: Same Thing?
This question is asked very often and creates confusion.
Olivine is mineral name. Peridot is name given to gem-quality form of this mineral.
So every peridot is olivine, but every olivine is not peridot. Peridots used as jewelry are generally more transparent, cleaner and shinier examples.
Olivine Physical Properties
Olivine is mineral of medium hardness. Neither extremely fragile nor very durable. Has glass shine and generally gives oily surface feeling.
Doesn’t show cleavage, which makes it more predictable during cutting. However if there are internal cracks needs to be processed carefully.
Color and shine make olivine visually attractive but real value of this mineral is in its geological meaning.
| Chemical Classification | Silicate |
| Color | Usually olive green, but can be yellow-green to bright green; iron-rich specimens are brownish green to brown |
| Streak | Colorless |
| Luster | Vitreous |
| Diaphaneity | Transparent to translucent |
| Cleavage | Poor cleavage, brittle with conchoidal fracture |
| Mohs Hardness | 6.5 to 7 |
| Specific Gravity | 3.2 to 4.4 |
| Diagnostic Properties | Green color, vitreous luster, conchoidal fracture, granular texture |
| Chemical Composition | Typically (Mg, Fe)2SiO4. Ca, Mn, and Ni rarely occupy the Mg and Fe positions. |
| Crystal System | Orthorhombic |
| Uses | Gemstones, a declining use in bricks and refractory sand |
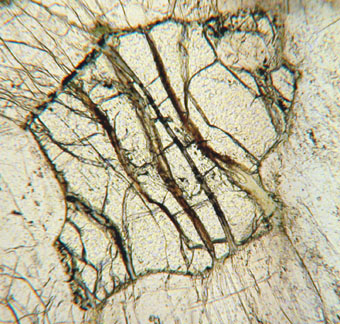
Olivine Optical Properties
| Formula | (MgFe)2SiO4 |
| Crystal System | Orthorhombic |
| Crystal Habit | Granular masses or rounded grains |
| Cleavage | Poor cleavage on (010) and (110) |
| Color/Pleochroism | Olive or yellowish-green in hand samples. Colorless to pale green in thin section. Weak, pale green pleochroism in thin section. |
| Optic Sign | Biaxial (-); or Biaxial (+) |
| 2V | 82-90; forsterite 46-90; fayalite |
| Optic Orientation | X=b Y=c Z=a O.A.P. = (001) |
| Refractive Indices alpha = beta = gamma = delta = | forsterite-fayalite 1.635-1.827 1.651-1.869 1.670-1.879 0.035-0.052 |
| Extinction | parallel |
| Dispersion | Relatively weak |
| Distinguishing Features | Olivine is commonly recognized by it high retardation, distinctive fracturing, lack of cleavage, and alteration to serpentine. Colorless to olive green in thin section. Second-order interference colors. High relief. Lack of cleavage. H= 7. G = 3.22 to 4.39. Specific gravity increases and hardness decreases with increasing Fe. Streak is colorless or white. |
| Sources | Nesse (1986) Introduction to Optical Mineralogy. Mindat.org. |
Where Is Olivine Used?
Most known use of Olivine is in jewelry sector as peridot. However apart from this has important usage areas.
In industry, can be used in refractory materials thanks to its high temperature resistant structure. Also olivine sands are preferred in metallurgy and casting industry.
In geology olivine is one of key minerals for understanding Earth’s internal structure. Is located at center of subjects like mantle composition, plate tectonics and volcanism.
What Makes Olivine So Special?
Olivine doesn’t shout like showcase stone. But its story is very deep. This mineral is window opening to internal structure of our planet.
Small green crystal in your hand actually came from kilometers below Earth. And this thought makes olivine much more than ordinary green stone.
References
- Bonewitz, R. (2012). Rocks and minerals. 2nd ed. London: DK Publishing.
- Dana, J. D. (1864). Manual of Mineralogy… Wiley.
- Mindat.org. (2019): Mineral information, data and localities.. [online] Available at: https://www.mindat.org/ [Accessed. 2019].
- Smith.edu. (2019). Geosciences | Smith College. [online] Available at: https://www.smith.edu/academics/geosciences [Accessed 15 Mar. 2019].