When you look at a crystal, the first thing you usually notice is its shape.
Flat faces, sharp edges, repeating geometries.
It looks like someone sat down and drew it with a ruler.
But the interesting part is this:
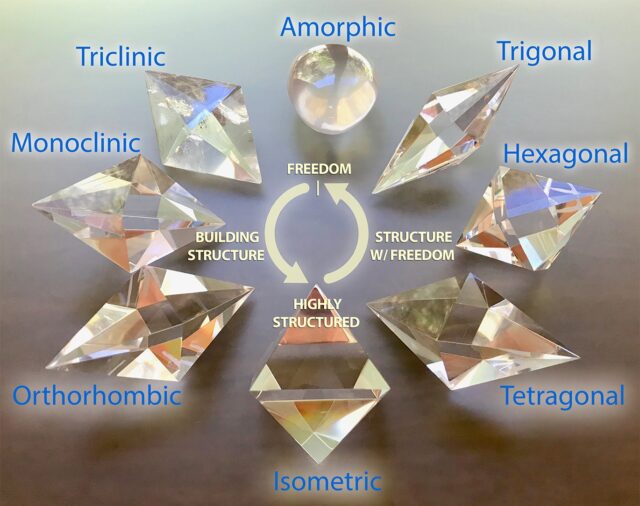
All crystals found in nature, no matter how different they look, actually belong to only seven basic crystal systems.
Diamond.
Salt crystals.
Quartz.
Emerald.
All of them grow according to the rules of one of these seven systems.
In this article, we will explain step by step what crystal systems are, why there are only seven of them, and how these systems determine the shape of minerals.
What is a crystal system?
A crystal system describes how a crystal is organized at the atomic scale.
In other words:
- How atoms are arranged in space
- Along which axes they repeat
- At what angles these axes are connected to each other
The important point is this:
A crystal system is not the shape you see from the outside.
It is the internal order behind that shape.
Because of this, two minerals:
- Can look similar from the outside
- But belong to completely different crystal systems
Or the opposite:
- They can belong to the same crystal system
- But show very different shapes
Are crystal system and crystal shape the same thing?
No. And this is where confusion happens most often.
- Crystal system → internal atomic arrangement
- Crystal habit → the external shape of the crystal
For example:
- Quartz usually looks like a hexagonal prism
- But this is its habit
- What really matters is that its atoms are arranged in the trigonal system
So you cannot always correctly guess the crystal system just by looking at the shape.
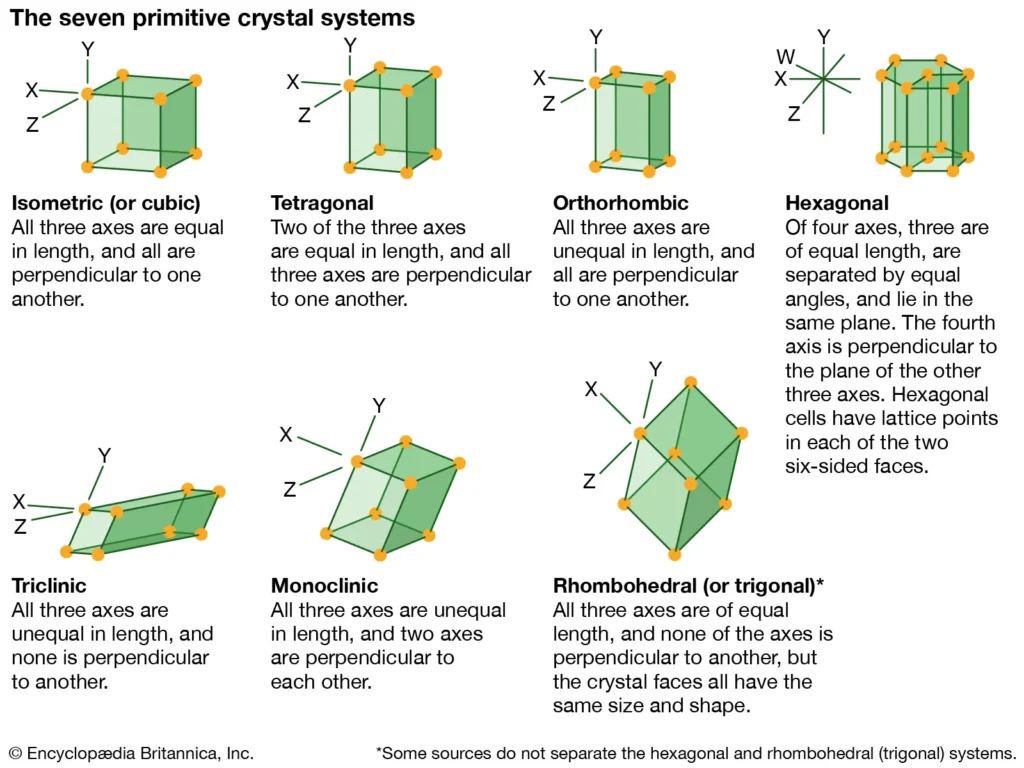
Why are there only 7 crystal systems?
Because atoms cannot arrange themselves in space in unlimited ways, but only according to certain mathematical rules.
Crystallography classifies these arrangements based on:
- The number of axes
- The lengths of the axes
- The angles between the axes
At the end of this classification, seven basic systems that can form stably in nature appear.
These are:
- Cubic (Isometric)
- Tetragonal
- Hexagonal
- Trigonal
- Orthorhombic
- Monoclinic
- Triclinic
Now let’s examine them one by one, in a simple way.
1. Cubic (Isometric) Crystal System

This system is the most symmetrical.
Its properties:
- There are three axes
- All of them are equal in length
- They intersect each other at 90 degrees
Because of this, crystals in this system usually look like:
- Cubes
- Octahedrons
- Dodecahedrons
Common minerals:
- Halite (rock salt)
- Galena
- Pyrite
- Diamond
The cubic breaking of halite crystals is not a coincidence.
The atomic arrangement makes this necessary.
2. Tetragonal Crystal System

This system is similar to the cubic system, but there is a small difference.
Its properties:
- There are three axes
- Two are equal in length
- The third axis is a different length
- All angles are 90 degrees
This gives crystals:
- An elongated prismatic shape
- Growth mainly in the vertical direction
Common minerals:
- Zircon
- Rutile
- Cassiterite
The needle-like elongation of rutile crystals is related to this system.
3. Hexagonal Crystal System

As the name suggests, this system has hexagonal symmetry.
Its properties:
- There are four axes
- Three are in the same plane and at 120-degree angles
- The fourth axis is perpendicular to them
This structure causes crystals to grow as:
- Hexagonal prisms
- Layered structures
Common minerals:
- Beryl (emerald, aquamarine)
- Apatite
- Graphite
The characteristic hexagonal prism shape of emerald is a direct result of this system.
4. Trigonal Crystal System

The trigonal system is often confused with the hexagonal system.
But they are not the same.
Its properties:
- The symmetry is threefold
- Even if the structure looks hexagonal, the atomic arrangement is different
This system can create:
- Spiral-like symmetry
- Inclined crystal faces
Common minerals:
- Quartz
- Calcite
- Corundum
The key to understanding why quartz looks hexagonal but is not classified as hexagonal lies here.
5. Orthorhombic Crystal System

In this system, symmetry is lower.
Its properties:
- There are three axes
- All have different lengths
- But the angles are still 90 degrees
This gives crystals:
- An asymmetric but ordered structure
- A rectangular appearance
Common minerals:
- Olivine
- Sulfur
- Topaz
The “irregular but balanced” look of olivine crystals comes from this system.
6. Monoclinic Crystal System

Here, symmetry decreases even more.
Its properties:
- There are three axes
- Two angles are 90 degrees
- The third angle is inclined
This causes crystals to look:
- Tilted
- Shifted
Common minerals:
- Gypsum
- Orthoclase feldspar
- Malachite
This is where you understand why gypsum crystals often look “crooked.”
7. Triclinic Crystal System

This is the system with the least symmetry.
Its properties:
- There are three axes
- None of them are equal
- None of the angles are 90 degrees
In this system, crystals look:
- Completely asymmetric
- Complex
Common minerals:
- Albite
- Kyanite
- Turquoise
Now it is clear why these minerals look “ruleless.”
Why are crystal systems important?
Because the crystal system directly affects:
- Physical properties of the mineral
- Cleavage and fracture
- Optical behavior
- Mechanical strength
For this reason, crystal systems are basic knowledge in:
- Gemology
- Petrography
- Materials science
- Industrial mineral use
Conclusion
Crystals do not grow randomly.
Each one is:
- Defined at the atomic scale
- Mathematically structured
- Bound by physical rules
The seven crystal systems are the structural framework behind all this diversity in nature.
When you look at a crystal, you no longer see only its shape.
You also see the internal order behind it.




























