(How Geologists Identify and Classify Minerals in the Field)
Understanding the physical properties of minerals is one of the most fundamental skills in geology. Every mineral has a set of characteristics that make it unique — its own “fingerprint” formed deep within the Earth. By observing and testing these properties, geologists can determine a mineral’s identity, its origin, and even its potential industrial or gemological value.
Physical properties are the visible and measurable expressions of a mineral’s chemical composition and atomic structure. They help explain why some minerals sparkle, some fracture easily, and others can cut through steel.
1. Color and Appearance
Color is the most noticeable property, yet often the most misleading. Many minerals show a wide range of colors due to impurities or exposure to light and heat. For instance, quartz may appear colorless, pink (rose quartz), or purple (amethyst) depending on trace elements and radiation effects.
Geologists usually use color only as a supporting clue, not a defining factor. More reliable observations include transparency, surface reflection, and crystal habit — the typical shape in which the mineral grows.

2. Luster
Luster describes how light interacts with the surface of a mineral. It can be metallic, vitreous (glassy), pearly, silky, or dull, depending on the atomic arrangement and surface texture.
- Metallic luster (e.g., pyrite, galena) reflects light like polished metal.
- Vitreous luster (e.g., quartz, calcite) resembles the shine of glass.
- Pearly or silky luster (e.g., talc, gypsum) often appears on layered or fibrous surfaces.
This property is crucial when classifying opaque minerals in ore deposits.

3. Streak
The streak is the color of a mineral’s powder, obtained by rubbing it across an unglazed porcelain plate. Unlike surface color, streak is consistent and unaffected by weathering.
For example:
- Hematite shows a reddish-brown streak, even if the crystal appears silver or black.
- Magnetite always leaves a dark gray streak.
Streak helps geologists distinguish between minerals that look identical in hand specimens.

4. Hardness
The hardness of a mineral is its resistance to scratching. It is measured using Mohs scale, which ranges from 1 (talc) to 10 (diamond). Each step in the scale can scratch all minerals below it.
| Mohs Hardness | Example Mineral |
|---|---|
| 1 | Talc |
| 2 | Gypsum |
| 3 | Calcite |
| 4 | Fluorite |
| 5 | Apatite |
| 6 | Orthoclase feldspar |
| 7 | Quartz |
| 8 | Topaz |
| 9 | Corundum |
| 10 | Diamond |
Hardness tests are essential in gemology, construction material testing, and geotechnical engineering, where durability and wear resistance matter.

5. Cleavage and Fracture

Cleavage describes how a mineral splits along specific planes of weakness — flat surfaces where atomic bonds are weakest. Minerals like mica show perfect cleavage, breaking into thin sheets.
Fracture, on the other hand, occurs when a mineral breaks irregularly without planar surfaces. Quartz typically exhibits a conchoidal fracture, creating curved, shell-like surfaces.
Understanding cleavage and fracture helps identify minerals under both field and microscopic conditions, and reveals insights into their crystal structure.
6. Density (Specific Gravity)
Density, or specific gravity, indicates how heavy a mineral feels for its size. It’s defined as the ratio of the mineral’s mass to an equal volume of water.
- Light minerals (e.g., quartz, feldspar) have a specific gravity around 2.5–2.8.
- Heavy minerals (e.g., galena, magnetite) may exceed 5.0.
This property is important in ore geology and mineral separation techniques used in mining and gem processing.

7. Magnetism
Some minerals exhibit magnetic properties due to iron, cobalt, or nickel content. The most famous example is magnetite, which strongly attracts a magnet.
Magnetism is not only a useful identification tool but also a key factor in understanding the mineral’s chemical composition and geologic environment. In exploration geology, magnetic data often help locate iron-rich ore bodies.

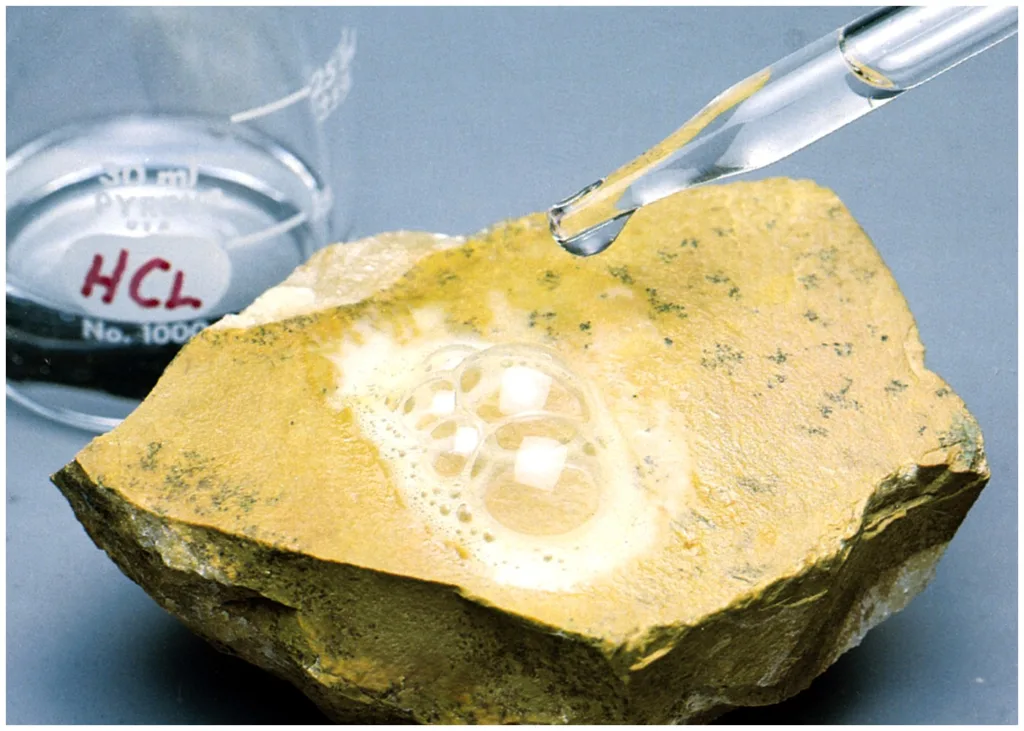
8. Reaction to Acid
Certain carbonate minerals, like calcite or dolomite, react with dilute hydrochloric acid (HCl), producing carbon dioxide bubbles.
This simple test distinguishes carbonates from silicates or oxides and is widely used in both laboratory and fieldwork.
9. Other Diagnostic Properties
Some minerals display additional, distinctive properties:
- Fluorescence under UV light (fluorite, scheelite).
- Taste or odor (halite tastes salty, sulfur smells like rotten eggs).
- Feel (talc feels greasy or soapy).
- Tenacity — how a mineral responds to stress: brittle, malleable, elastic, or flexible.
Though secondary, these properties often confirm identification results obtained through primary tests.

10. Why Physical Properties Matter
Understanding physical properties is not limited to academic curiosity. These traits influence how minerals are mined, processed, and used in industries ranging from construction to jewelry.
- Engineers assess hardness and density to choose materials for building and road construction.
- Gemologists evaluate luster, cleavage, and color for gemstone cutting.
- Environmental scientists study mineral reactions to predict soil and groundwater chemistry.
By mastering these properties, geologists connect what they observe on the surface with what happens deep inside the Earth — linking appearance, structure, and origin into one scientific story.
? Summary Table
| Property | Description | Example |
|---|---|---|
| Color | Visual appearance, often variable | Quartz, feldspar |
| Luster | How light reflects from surface | Pyrite (metallic), quartz (vitreous) |
| Streak | Powder color | Hematite (red-brown) |
| Hardness | Resistance to scratching | Diamond (10) |
| Cleavage/Fracture | Breakage pattern | Mica (cleavage), quartz (fracture) |
| Density | Weight per volume | Galena (7.5 SG) |
| Magnetism | Magnetic response | Magnetite |
| Acid Reaction | Effervescence with HCl | Calcite |
| Others | Fluorescence, taste, tenacity | Fluorite, halite |
Conclusion
The study of mineral physical properties bridges observation and science. Each property — whether color, luster, or density — tells part of the story of Earth’s formation. By learning to interpret them, we not only identify minerals but also understand the conditions that created them.