You pick up a stone. You look at it in daylight.
It looks ordinary. Gray, white, maybe slightly colored.
For most people, the story ends here.
Then someone turns on a UV lamp.
The stone suddenly starts to glow. It becomes green, blue, orange.
Sometimes it is so bright that your eyes automatically focus on it.
The first reaction is usually the same:
“Is this real?”
Yes, it is real.
And there is no magic and no trick behind it.
This is completely a point where geology, chemistry, and physics come together.
This is where fluorescent minerals come in.
What is a fluorescent mineral?

A fluorescent mineral is a mineral that emits visible light under ultraviolet (UV) light.
In other words:
- It absorbs a type of light that we normally cannot see
- It takes this energy
- And gives it back at a different wavelength
- As colors that our eyes can see
There is a very important difference here:
- It glows when the UV light is on
- The glow stops when the UV light is turned off
For this reason, fluorescence is not the same as phosphorescence.
Fluorescence = while the light is on
Phosphorescence = continues even after the light is off
Fluorescent minerals are common in nature.
True phosphorescence, however, is quite rare.
How does this glowing happen?

The reason a mineral shows fluorescence is the presence of activator elements inside its crystal structure.
These elements are not the main components of the mineral.
They exist in very small amounts, but their effect is large.
The most common activator elements are:
- Manganese (Mn)
- Europium (Eu)
- Lead (Pb)
- Terbium (Tb)
- Uranium (U)
For example:
- Calcite is calcium carbonate
- But very small amounts of manganese inside it
- Can cause red or orange glowing under UV light
The process works in a simple way:
- UV light hits the mineral
- Electrons take energy and move to a higher energy level
- Electrons cannot stay at this level
- When they return, they release extra energy as light
This light is the color that we see.
What we are actually seeing is electrons returning to their original state.
Why is not every mineral fluorescent?
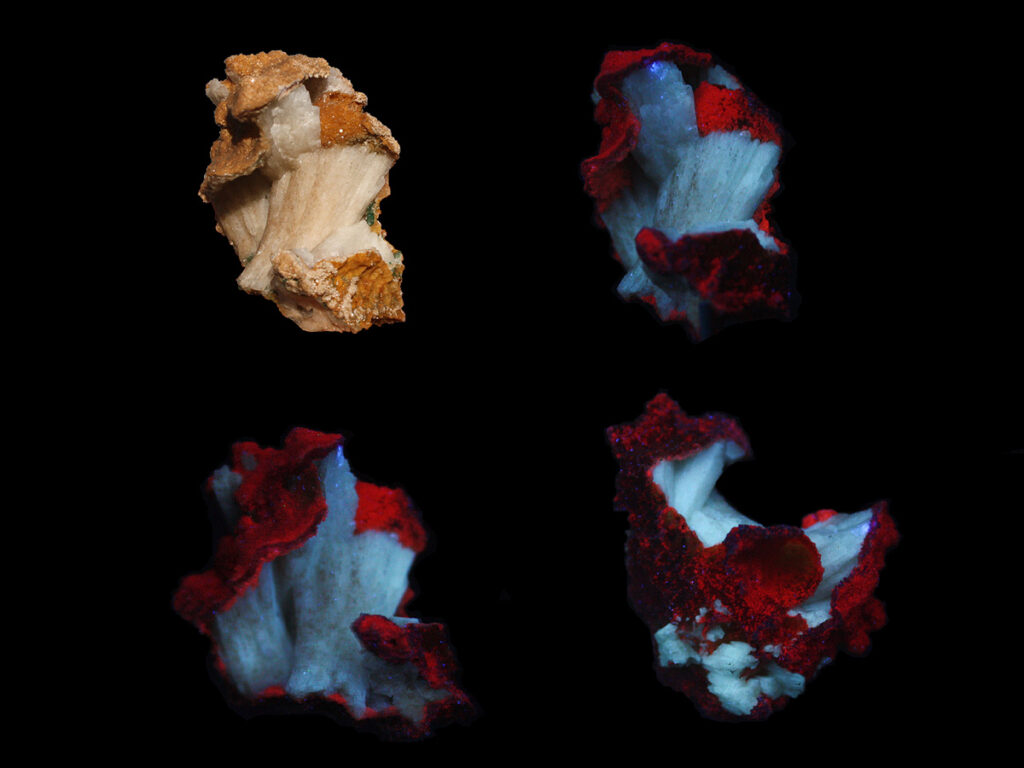
Fluorescent Minerals Of Africa Gallery
This question is asked very often.
The answer is simple but important.
Fluorescence requires more than one condition at the same time.
- There must be an activator element
- The crystal structure must allow this process
- There must not be other elements that block the effect
Some elements block fluorescence.
This is called the quenching effect.
Because of this:
- Two samples of the same mineral
- Even if they look the same
- Can behave completely differently under UV light
One glows.
The other does nothing.
This uncertainty is one of the reasons why fluorescent minerals are interesting.
Difference between shortwave UV and longwave UV

UV light is not a single type.
Longwave UV (LW – 365 nm)
- Safer
- Common in home UV lamps
- Effective for minerals like calcite and fluorite
Shortwave UV (SW – 254 nm)
- Stronger
- Used for professional purposes
- Produces more dramatic colors
- Must be used carefully
Some minerals:
- Glow only under shortwave UV
- Some glow only under longwave UV
- Some show different colors under both
This makes fluorescence more interesting.
Most well-known fluorescent minerals
Fluorite
Fluorescence is usually associated with fluorite.
- Blue
- Purple
- Green
- Yellow
The same crystal can show different colors under different UV wavelengths.
Calcite
Calcite is one of the most variable fluorescent minerals.
- Red
- Orange
- Pink
- Blue
The color depends completely on the trace elements inside the crystal.
Willemite
Willemite is known for its bright green fluorescence.
It is usually found together with franklinite and zincite.
Autunite
Autunite glows bright neon green because of its uranium content.
It is visually impressive but needs attention.
Yooperlite
Yooperlite is not a single mineral. It is a rock.
It glows under UV light because of fluorescent sodalite inside it.
This is the main reason for its popularity.
Fluorescence and crystal defects
Perfect crystals usually do not show fluorescence.
Fluorescence usually happens because of:
- Empty spaces in the crystal lattice
- Atomic level distortions
- Foreign ions replacing normal atoms
Geologically imperfect crystals are often more interesting under UV light.
Nature prefers imperfections.
Yes, it is directly related.
Fluorescent minerals are commonly found in:
- Hydrothermal vein systems
- Carbonate-rich rocks
- Certain metamorphic zones
Because of this, fluorescence can sometimes give information about the formation environment of a mineral.
What are fluorescent minerals used for in geology?

They are not used only for collecting.
In geology, fluorescence is used for:
- Mineral identification
- Carbonate rock analysis
- Mapping vein zones
- Uranium exploration
In some field studies, a UV lamp is as important as a rock hammer.
Can fluorescence be fake?
Yes.
Some stones are:
- Dyed
- Coated
- Treated with UV-reactive materials
Real fluorescent minerals:
- Usually look normal in daylight
- Glow only under UV light
- Show color coming from inside the mineral, not the surface
This difference is very important for collectors.
Why are fluorescent minerals so popular?
Because:
- They are visually striking
- They attract attention on social media
- They give a feeling of a hidden feature
- They make science more interesting
A stone that shows itself only under the right light has always attracted people.
Is every fluorescent mineral valuable?
No.
Fluorescence alone is not enough.
Value depends on:
- Rarity
- Crystal quality
- Size
- Visual appearance
However, strong and rare fluorescence can increase collector value.
Looking from a geological time perspective
When you look at a fluorescent mineral, it is useful to think about this:
- The stone formed millions of years ago
- The atoms inside it have been there since that time
- The reaction seen today is a result of those conditions
Fluorescence is the meeting of modern light and ancient chemistry.
Conclusion
Fluorescent minerals are not only glowing stones.
They are:
- The result of energy behavior at the atomic level
- Silent witnesses of geological history
- The point where chemistry, physics, and geology meet
When you look at a mineral under a UV lamp, what you see is not only color.
It is a small reflection of how that stone formed.