What is alteration?
Alteration refers to a change in the physical or chemical properties of rocks and minerals. In geology, alteration is a common term used to describe the transformation of rocks and minerals due to various geological processes, such as weathering, metamorphism, and hydrothermal activity.
For example, hydrothermal alteration occurs when hot, mineral-rich fluids interact with rocks and minerals, causing them to change in terms of their mineral composition, texture, and structure. The alteration of rocks and minerals can result in the formation of new minerals, and in some cases, the concentration of valuable minerals such as gold and silver.
In general, understanding the extent and nature of alteration is important for mineral exploration and mining, as it provides information about the location and type of minerals present in an area, and can help geologists and miners target areas for exploration and extraction.
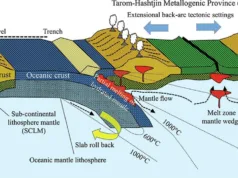
Hydrothermal alteration is a geological process that occurs when hot, mineral-rich fluids interact with rocks and minerals, changing their physical and chemical properties. This interaction can lead to the formation of new minerals and the alteration of existing minerals, which can result in the formation of mineral deposits, including those containing metals such as copper, gold, and silver.
Hydrothermal alteration can occur in a variety of geological settings, such as volcanic environments, hot springs, and geothermal systems. The fluids involved in hydrothermal alteration can be derived from magma or other deep sources, and can carry dissolved metals and minerals as they move through the Earth’s crust.
The extent and nature of hydrothermal alteration are important for mineral exploration and mining, as they provide valuable information about the location and type of minerals present in an area. By understanding the geological processes that led to the formation of mineral deposits, geologists and miners can better target areas for exploration and extraction.
Importance of Hydrothermal Alteration and Mineral Exploration
Hydrothermal alteration is important in mineral exploration and mining because it can provide valuable information about the location and type of minerals present in an area. By understanding the geological processes that led to the formation of mineral deposits, geologists and miners can better target areas for exploration and extraction.
For example, hydrothermal alteration can result in the formation of new minerals and the concentration of valuable minerals such as gold and silver. The extent and nature of hydrothermal alteration can indicate the presence of mineral deposits, and can provide information about the mineralization process and the conditions that existed at the time of mineral formation.
In addition, hydrothermal alteration can also affect the physical and chemical properties of rocks and minerals, making them easier or more difficult to extract. By understanding the extent and nature of alteration, miners can develop more effective extraction methods and minimize the impact of mining on the environment.
In summary, the importance of hydrothermal alteration in mineral exploration and mining lies in its ability to provide valuable information about the location, type, and characteristics of mineral deposits, and to inform effective exploration and extraction strategies.
- Feature of hydrothermal ore deposits
- Relates to type of deposit-environment
- Provides halo around target
- Vectors towards mineralization
Indication of size/intensity of system, may equate to potential The areal extent of the alteration can vary considerably, sometimes being limited to a few centimeters on either side of a vein, at other times forming a thick halo around an orebody
Controls of Alteration
There are several factors that control the extent and nature of hydrothermal alteration. Some of the key controls include:
- Temperature: The temperature of the hydrothermal fluids plays a major role in determining the extent and nature of alteration. Higher temperatures result in more intense alteration, while lower temperatures result in less intense alteration.
- Fluid Composition: The composition of the hydrothermal fluids can also influence the extent and nature of alteration. Different minerals will form depending on the composition of the fluids, so it is important to understand the composition of the fluids in order to predict the nature of the alteration.
- Pressure: The pressure of the hydrothermal fluids can affect the extent and nature of alteration. Higher pressures can result in more intense alteration, while lower pressures can result in less intense alteration.
- Fluid Flow: The flow of hydrothermal fluids through the rock is another important factor that controls the extent and nature of alteration. Faster fluid flow can result in more intense alteration, while slower fluid flow can result in less intense alteration.
- Host Rock: The type of host rock can also affect the extent and nature of alteration. Different types of rocks can have different permeabilities, and the permeability of the rock will influence the rate and extent of fluid flow and therefore the nature of the alteration.
- Time: The duration of hydrothermal fluid flow can also play a role in the extent and nature of alteration. Over time, more intense alteration can occur if the fluid flow is sustained.
By understanding the controls of hydrothermal alteration, geologists and miners can better predict the extent and nature of alteration, and therefore the location and type of mineral deposits.
Alteration intensity
Alteration intensity refers to the degree to which the host rock has been changed by hydrothermal fluid interactions. It is a measure of the extent of mineral replacement, mineral growth, and mineral dissolution that has occurred within the rock. High alteration intensity indicates a more extensive alteration event, while low alteration intensity suggests a more limited or shallow alteration event. The intensity of alteration can be an important factor in determining the potential for mineralization and the type of deposit that may have formed. In mineral exploration, the alteration intensity is usually evaluated based on the abundance and distribution of alteration minerals, the degree of homogenization or zoning within the altered rock, and the overall volume of altered rock compared to unaltered rock. The intensity of alteration can also vary within a single hydrothermal system, with some parts of the system experiencing higher alteration intensity than others.
Types of alterations
There are several types of hydrothermal alteration that can occur in geological systems, including:
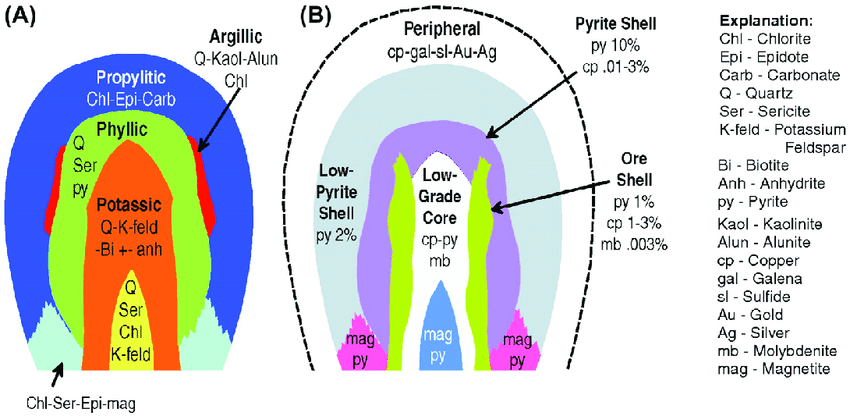
- Propylitic alteration: characterized by the formation of minerals such as chlorite, epidote, and sericite.
- Phyllic alteration: characterized by the formation of minerals such as muscovite, kaolinite, and sericite.
- Argillic alteration: characterized by the formation of minerals such as kaolinite, halloysite, and dickite.
- Silicic alteration: characterized by the formation of minerals such as quartz, silica, and chalcedony.
- Advanced argillic alteration: characterized by the formation of minerals such as pyrophyllite, diaspore, and kaolinite.
- Potassic alteration: characterized by the formation of minerals such as K-feldspar and biotite.
- Sodic alteration: characterized by the formation of minerals such as albite and nepheline.
The specific type of alteration that occurs can be influenced by a number of factors, including the chemical composition of the fluid, the temperature and pressure conditions, the host rock composition, and the duration and intensity of the fluid-rock interaction. Understanding the type of alteration that has occurred can be important in mineral exploration as it can provide clues as to the nature of the hydrothermal system and the type of mineralization that may be present.
Propylitic alteration

Propylitic alteration is a type of hydrothermal alteration that occurs in volcanic and plutonic rocks. It is characterized by the alteration of primary minerals, such as feldspar and quartz, to secondary minerals, such as chlorite, epidote, and sericite. Propylitic alteration typically occurs at lower temperatures (less than 200°C) and involves the introduction of hydrogen ions and other elements into the rock. This type of alteration is often associated with the formation of copper and gold deposits and is an important indicator of potential mineralization. In mineral exploration, propylitic alteration can be used as a guide to help identify areas with a higher likelihood of hosting mineral deposits.
Phyllic alteration

Phyllic alteration is a type of hydrothermal alteration that occurs at higher temperatures (typically between 200°C and 400°C) and is characterized by the alteration of primary minerals to secondary minerals such as muscovite, kaolinite, and sericite. Unlike propylitic alteration, phyllic alteration typically involves the removal of most of the original primary minerals and their replacement by secondary minerals. This type of alteration is often associated with the formation of porphyry copper and gold deposits and is an important indicator of potential mineralization. In mineral exploration, phyllic alteration can be used as a guide to help identify areas with a higher likelihood of hosting mineral deposits.
Argillic alteration

Argillic alteration is a type of hydrothermal alteration that occurs at even higher temperatures (typically greater than 400°C) and is characterized by the formation of clay minerals, such as illite and kaolinite, from the alteration of primary minerals such as feldspar and quartz. Argillic alteration typically occurs in the upper levels of a hydrothermal system, above the zone of phyllic alteration, and is often associated with porphyry copper and gold deposits. In addition to the formation of clay minerals, argillic alteration may also result in the formation of silica minerals, such as quartz and chalcedony, and the enrichment of certain elements, such as gold, silver, and molybdenum. The presence of argillic alteration is an important indicator of the potential for mineralization, and is often used in mineral exploration to help identify areas with a higher likelihood of hosting mineral deposits.
Silicic alteration

Tay Zar, Aung & Warmada, Iwayan & Setijadji, Lucas & Watanabe, Koichiro. (2017). Geochemical Characteristics of Metamorphic Rock-Hosted Gold Deposit At Onzon-Kanbani Area, Central Myanmar. Journal of Geoscience, Engineering, Environment, and Technology. 2. 191. 10.24273/jgeet.2017.2.3.410.
Silicic alteration is a type of hydrothermal alteration that results in the formation of silica minerals, such as quartz and chalcedony. It occurs at even higher temperatures (typically greater than 500°C) than argillic alteration and is typically found in the uppermost levels of a hydrothermal system. Silicic alteration is often associated with porphyry copper and gold deposits, as well as other types of mineral deposits. The formation of silica minerals during silicic alteration results in the destruction of primary minerals, such as feldspar, and the creation of a more silicic-rich rock. The presence of silicic alteration is an important indicator of a hydrothermal system, and is often used in mineral exploration to help identify areas with a higher likelihood of hosting mineral deposits.
Advanced argillic alteration
Advanced argillic alteration is a type of hydrothermal alteration that results in the formation of clay minerals, such as kaolinite and dickite. It is typically found in the deeper levels of a hydrothermal system and occurs at higher temperatures than propylitic alteration. Advanced argillic alteration is characterized by the destruction of primary minerals, such as feldspar and mica, and the formation of clay minerals. The presence of advanced argillic alteration is often used as an indicator of a mineral deposit, particularly in the case of porphyry copper and gold deposits. The clay minerals formed during advanced argillic alteration can also act as a host for other minerals, such as gold and copper, making the alteration zone a potential target for exploration.
Potassic alteration or Potassium silicate alteration
Potassic alteration is a type of hydrothermal alteration that results in the formation of potassium-rich minerals, such as orthoclase, sanidine, and microcline. This type of alteration is typically associated with porphyry copper and gold deposits and is considered an important mineralization indicator. Potassic alteration occurs at intermediate to high temperatures and is characterized by the replacement of primary minerals, such as plagioclase and biotite, with potassium-rich minerals. Potassic alteration can also result in the formation of biotite and muscovite, which are important indicators of the intensity of alteration. The potassium-rich minerals formed during potassic alteration can also act as a host for other minerals, such as molybdenum and gold, making the alteration zone a potential target for exploration. The style and intensity of potassic alteration can vary greatly depending on the specific geologic setting and hydrothermal conditions.

Sodic alteration

Sodic alteration refers to the type of hydrothermal alteration that results from the introduction of sodium into the host rock. This type of alteration is typically characterized by the presence of minerals such as albite, potassium feldspar, and sanidine. Sodic alteration is often associated with porphyry copper deposits and is often accompanied by other types of alteration such as phyllic and argillic alteration. The style and intensity of sodic alteration can provide important information for mineral exploration and the understanding of the mineralizing processes that took place during ore formation.