Carbonate Replacement Deposits (CRDs) are geological formations that result from the replacement of pre-existing carbonate rocks by ore minerals, often metals such as lead, zinc, and copper. These deposits are significant sources of base metals and are of economic importance due to the concentration of valuable minerals within them.

Basic Characteristics:
- Formation Process: CRDs typically form through a replacement process where hydrothermal fluids rich in metals percolate through carbonate rocks, dissolving the original minerals and replacing them with ore minerals. The replacement process occurs in response to changes in temperature, pressure, and chemical composition of the fluids.
- Ore Minerals: The primary ore minerals found in CRDs include sphalerite (zinc), galena (lead), and chalcopyrite (copper). These minerals often accumulate within the altered carbonate host rocks, creating economically viable deposits.
- Host Rocks: The host rocks for CRDs are carbonate rocks such as limestone and dolomite. The replacement of these carbonate rocks by ore minerals leads to the formation of distinct mineralized zones within the deposit.
- Spatial Distribution: CRDs can exhibit a wide range of spatial distributions, from localized ore bodies to extensive mineralized zones. The distribution of ore minerals is influenced by geological structures, fluid pathways, and the nature of the host rocks.
Historical Context and Discovery: The discovery of CRDs dates back to the late 19th and early 20th centuries. One of the notable early discoveries occurred in the famous Broken Hill deposit in Australia in 1883. Broken Hill is a classic example of a CRD, with lead, zinc, and silver minerals replacing carbonate rocks.
Over time, CRDs have been identified in various geological settings around the world. Mexico, the United States, Canada, Peru, and China are among the countries that host significant CRD deposits. Advances in geological understanding and exploration techniques have played a crucial role in the continued discovery of CRDs.
Importance: CRDs are economically important as they can host high concentrations of valuable metals. The mining of these deposits contributes significantly to the global production of lead, zinc, and copper. Understanding the geological processes and characteristics of CRDs is essential for successful exploration and exploitation of these mineral resources.
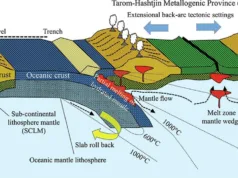
Geological Setting and Formation
Host Rocks: Carbonate Replacement Deposits (CRDs) primarily occur in carbonate sequences, with limestone and dolomite being the predominant host rocks. These carbonate rocks provide the necessary framework for the formation of CRDs through the replacement of original minerals by ore minerals.
Tectonic Settings Conducive to CRD Formation: CRDs are often associated with specific tectonic settings and geological environments. Some of the common tectonic settings conducive to CRD formation include:
- Folded Mountain Belts: CRDs are frequently found in regions associated with folded mountain belts. The compression and deformation associated with the tectonic activity in these settings create fractures and faults, providing pathways for hydrothermal fluids.
- Subduction Zones: Tectonic environments where one tectonic plate is subducting beneath another can be conducive to CRD formation. Subduction-related magmatism and fluid circulation can lead to the alteration and replacement of carbonate rocks.
- Rift Zones: Rift zones, where the Earth’s lithosphere is being pulled apart, can create favorable conditions for the circulation of hydrothermal fluids. The extensional tectonics associated with rift zones can result in the development of fractures and faults, providing pathways for mineralizing fluids.
- Fault Zones: Fault systems, regardless of the specific tectonic setting, can play a crucial role in CRD formation. Faults act as conduits for hydrothermal fluids, allowing them to migrate through the Earth’s crust and interact with carbonate rocks.
Hydrothermal Processes Involved in CRD Formation: The formation of Carbonate Replacement Deposits involves complex hydrothermal processes. Here are the key steps:
- Hydrothermal Fluids: Hot, metal-rich fluids, often associated with magmatic activity, circulate through the Earth’s crust. These fluids may originate from the mantle or from deeper parts of the crust.
- Fluid-Rock Interaction: The hydrothermal fluids interact with the carbonate host rocks (limestone and dolomite). This interaction involves the dissolution of original carbonate minerals and the precipitation of ore minerals in their place. The replacement process is driven by changes in temperature, pressure, and chemical composition of the fluids.
- Zoning: CRDs often exhibit a zonal pattern, with different mineralization zones corresponding to variations in temperature, pressure, and fluid composition. This zoning can include central zones with the highest metal concentrations surrounded by peripheral zones with lower concentrations.
- Fracture and Fault-Related Mineralization: Faults and fractures within the host rocks provide conduits for the hydrothermal fluids. Mineralization is often concentrated along these structures, resulting in the formation of ore bodies within the broader CRD system.
Understanding the geological and hydrothermal processes involved in CRD formation is essential for mineral exploration and resource assessment. Advances in geological mapping, geochemistry, and geophysics contribute to the identification and characterization of potential CRD deposits.
Ore Minerals and Mineralization

Ore Minerals:
The primary ore minerals associated with Carbonate Replacement Deposits (CRDs) include:
- Sphalerite (Zinc Sulfide): Sphalerite is a commonly occurring ore mineral in CRDs and is the primary source of zinc. It often forms well-defined crystals and can vary in color from yellow to brown to black.
- Galena (Lead Sulfide): Galena is another significant ore mineral found in CRDs, serving as the primary source of lead. It typically appears as shiny, metallic cubes or octahedral crystals.
- Chalcopyrite (Copper Iron Sulfide): Chalcopyrite is a copper-bearing ore mineral present in some CRDs. It has a brassy yellow color and is an important source of copper.
- Tetrahedrite (Copper Antimony Sulfide): Tetrahedrite is sometimes found in CRDs, contributing to the copper content. It often occurs as dark, metallic crystals.
- Pyrite (Iron Sulfide): While pyrite is not a primary economic ore mineral in CRDs, it is often associated with the ore bodies. Pyrite forms cubic crystals and can be present in varying amounts.
Gangue Minerals:
Gangue minerals are non-economic minerals that are associated with ore deposits. In the case of CRDs, the following gangue minerals may be present:
- Calcite: Calcite is a common gangue mineral in CRDs, especially considering the carbonate host rocks. It often forms rhombohedral crystals and can be found intergrown with ore minerals.
- Dolomite: Dolomite, another carbonate mineral, can also be present as gangue in CRDs. It has a similar appearance to calcite but can be distinguished by its chemical composition.
- Quartz: Quartz is a common gangue mineral in many ore deposits, and it may be associated with CRDs. It forms hexagonal crystals and is resistant to weathering.
- Barite: Barite is occasionally found as a gangue mineral in CRDs. It has a high specific gravity and may form tabular crystals.
Textures and Paragenesis of Ore Minerals:
- Replacement Textures: The most characteristic texture in CRDs is replacement, where the original carbonate minerals are replaced by ore minerals. This replacement can occur with a preservation of the original rock fabric, leading to distinctive textures.
- Zoning: CRDs often exhibit zoning in mineralization, with different mineral assemblages corresponding to changes in temperature, pressure, and fluid composition. This zoning can include a central core of higher-grade ore minerals surrounded by peripheral zones with lower concentrations.
- Paragenesis: The paragenetic sequence in CRDs refers to the chronological order of mineral formation. It helps in understanding the evolution of the deposit over time. Typically, sulfide minerals like sphalerite and galena form early in the paragenetic sequence, followed by later-stage minerals like quartz and calcite.
- Crosscutting Veins: In addition to replacement, ore minerals in CRDs can form crosscutting veins within the host rocks. These veins are often associated with fractures and faults, representing later-stage mineralization events.
Understanding these ore minerals, gangue minerals, textures, and paragenetic relationships is crucial for both exploration and exploitation of CRDs. Geological studies, including detailed fieldwork and laboratory analyses, contribute to unraveling the complex history of these deposits.
Geochemical Signature of CRDs
The geochemical signature of Carbonate Replacement Deposits (CRDs) provides valuable information about the origin and evolution of the mineralizing fluids. Key geochemical indicators include:
- Metal Content: Elevated concentrations of metals such as zinc, lead, and copper are primary indicators of CRDs. Geochemical analyses of rock samples can reveal the presence of these economically valuable metals.
- Pathfinder Elements: Certain elements are associated with specific types of ore deposits. In the case of CRDs, pathfinder elements may include elements like silver, antimony, arsenic, and bismuth. These elements can serve as indicators during exploration.
- Sulfur Isotopes: The sulfur isotopic composition of sulfide minerals in CRDs can provide insights into the source of sulfur in the mineralizing fluids. Variations in sulfur isotopes may indicate contributions from different sources, such as magmatic or sedimentary sulfur.
- Carbon and Oxygen Isotopes: Carbonate minerals in CRDs, such as calcite and dolomite, can exhibit variations in carbon and oxygen isotopes. Isotopic studies help in understanding the source of carbon and oxygen in the hydrothermal fluids and can provide information about fluid-rock interaction.
Fluid Inclusion Studies:
Fluid inclusions are microscopic cavities within minerals that contain trapped fluids, providing direct evidence of the composition and characteristics of the mineralizing fluids. Fluid inclusion studies in CRDs involve:
- Fluid Composition: Analyzing the composition of fluids trapped in inclusions helps identify the chemical characteristics of the hydrothermal fluids responsible for mineralization.
- Temperature and Pressure Conditions: The study of fluid inclusions allows geologists to estimate the temperature and pressure conditions during mineralization. This information aids in reconstructing the geological history of the deposit.
- Salinity: The salinity of fluid inclusions is a crucial parameter. Changes in salinity can indicate variations in the chemical composition of the hydrothermal fluids during the evolution of the deposit.
- Phase Changes: Observing phase changes (e.g., vapor-liquid or liquid-liquid transitions) in fluid inclusions helps in determining the trapping conditions and understanding the fluid’s behavior.
Isotope Studies:
Isotope studies provide additional insights into the sources and processes involved in CRD formation:
- Stable Isotopes (Oxygen, Carbon): Stable isotopes of oxygen and carbon in carbonate minerals can indicate the temperature and source of the hydrothermal fluids. Variations in stable isotopes can help distinguish between different fluid sources and provide information on fluid-rock interaction.
- Radiogenic Isotopes (Lead, Strontium): Radiogenic isotopes, such as lead and strontium isotopes, can be used to establish the age of the mineralization and trace the origin of the metals. Isotope ratios help distinguish between different geological sources for the metals.
- Sulfur Isotopes: As mentioned earlier, sulfur isotopes in sulfide minerals provide information on the source of sulfur in the hydrothermal fluids.
Integration of these geochemical, fluid inclusion, and isotope studies allows geologists to build a comprehensive understanding of the genesis and evolution of CRDs, aiding in mineral exploration and resource assessment.
Types of Carbonate Replacement Deposits
Carbonate Replacement Deposits (CRDs) can exhibit various types and classifications based on their geological characteristics, mineralogy, and geological settings. Some common types of CRDs include:
- Mississippi Valley Type (MVT) Deposits:
- Irish-Type Zinc-Lead Deposits:
- Host Rock: Hosted in Carboniferous limestone.
- Minerals: Characterized by sphalerite and galena as primary ore minerals.
- Distribution: Found in Ireland and parts of the United Kingdom.
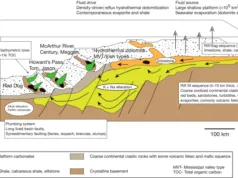
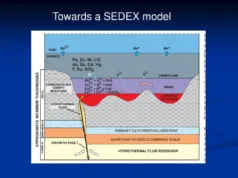
- SEDEX (Sedimentary Exhalative) Deposits:
- Host Rock: Hosted in sedimentary rocks, including carbonate sequences.
- Minerals: Composed of sulfide minerals such as sphalerite, galena, and pyrite. Barite may also be present.
- Distribution: Widely distributed globally, often associated with basins and rift settings.
- Broken Hill Type Deposits:
- Host Rock: Primarily hosted in carbonate rocks.
- Minerals: Characterized by galena, sphalerite, and minor amounts of other sulfides.
- Distribution: Notable examples include the Broken Hill deposit in Australia.
- Skarn-Type Deposits:
- Host Rock: Carbonate rocks that undergo metasomatic alteration due to intrusions of magmatic rocks.
- Minerals: Ore minerals include sphalerite, galena, and chalcopyrite, often associated with skarn minerals such as garnet and pyroxene.
- Distribution: Associated with contact metamorphism zones around intrusive igneous bodies.
- Strata-Bound Replacement Deposits:
- Host Rock: Typically occur in carbonate sequences within sedimentary basins.
- Minerals: Ore minerals can include sphalerite, galena, and other sulfides.
- Distribution: Found in stratigraphic horizons and can be influenced by regional tectonics.
- Hydrothermal Dolomite-Hosted Deposits:
- Host Rock: Dominantly hosted in dolomite.
- Minerals: Ore minerals such as sphalerite and galena are associated with dolomite replacement.
- Distribution: Occur in regions where dolomitization has taken place, often associated with hydrothermal fluid flow.
- Carbonate-Hosted Lead-Zinc (CHZ) Deposits:
- Host Rock: Carbonate rocks, including limestone and dolomite.
- Minerals: Mainly composed of galena and sphalerite.
- Distribution: Found in various geological settings, including platform carbonates and rift-related settings.
These types of CRDs demonstrate the diversity of geological environments and processes that can lead to the formation of economically significant mineral deposits. Each type has its own set of characteristics, and understanding these variations is crucial for successful mineral exploration and exploitation.
Regional Examples of CRDs
- Broken Hill Deposit, Australia:
- Location: New South Wales, Australia.
- Minerals: Predominantly galena (lead) and sphalerite (zinc).
- Geological Characteristics: Broken Hill is one of the world’s richest CRDs, with mineralization occurring in a sequence of Silurian sedimentary rocks. The deposit is associated with faulting and is hosted in a carbonate-rich environment. It has been a historically significant source of lead, zinc, and silver.
- Trepča Mines, Kosovo:
- Location: Northern Kosovo.
- Minerals: Galena, sphalerite, chalcopyrite, and pyrite.
- Geological Characteristics: The Trepča Mines represent a complex of CRDs hosted in carbonate rocks. The mineralization is associated with fault zones and occurs within a tectonically active region. The deposit has been historically important for lead, zinc, and other base metals.
- Pine Point Mine, Canada:
- Location: Northwest Territories, Canada.
- Minerals: Sphalerite, galena, and pyrite.
- Geological Characteristics: Pine Point is a classic example of a Mississippi Valley Type (MVT) deposit. The ore occurs in dolostone and limestone, and the mineralization is associated with karst features and faults. It was a significant lead-zinc producer in the past.
- Borieva Mine, Bulgaria:
- Location: Madan ore field, Bulgaria.
- Minerals: Sphalerite, galena, pyrite, and chalcopyrite.
- Geological Characteristics: The Borieva Mine is situated in a region with a long history of mining and is known for its carbonate-hosted ore deposits. The mineralization is associated with faulting and occurs within carbonate rocks, contributing to Bulgaria’s lead and zinc production.
- Rammelsberg Mine, Germany:
- Location: Lower Saxony, Germany.
- Minerals: Sphalerite, galena, pyrite, and chalcopyrite.
- Geological Characteristics: Rammelsberg is a historic mining district that has been exploited for centuries. The ore occurs in a polymetallic deposit hosted in a complex of volcanic and sedimentary rocks. It is one of the largest lead-zinc-silver deposits in the world.
- Ozdag Mining District, Turkey:
- Location: Central Anatolia, Turkey.
- Minerals: Sphalerite, galena, and pyrite.
- Geological Characteristics: The Ozdag Mining District is known for its carbonate-hosted CRDs. The mineralization is associated with fault zones, and the ore occurs in dolomite and limestone. Turkey has been a significant producer of zinc and lead from such deposits.
- Navan Mining District, Ireland:
- Location: County Meath, Ireland.
- Minerals: Sphalerite, galena, and pyrite.
- Geological Characteristics: The Navan Mining District is an Irish-type zinc-lead deposit. The ore occurs in Carboniferous limestone and is associated with faulting. It has been a major source of zinc and lead in Ireland.
These regional examples highlight the global distribution of Carbonate Replacement Deposits and the geological diversity of the environments in which they form. Each deposit has unique characteristics shaped by its geological history and tectonic setting, contributing to the economic significance of the respective mining districts.
Comparisons with Other Deposit Types
1. Porphyry Copper Deposits:
- Contrast: Porphyry copper deposits are primarily associated with magmatic intrusions and are characterized by disseminated mineralization in large volumes of host rock. In contrast, CRDs are typically hosted in carbonate rocks and result from the replacement of original minerals by ore minerals due to hydrothermal fluids.
- Commonality: Both deposit types can be significant sources of base metals, including copper, and are often associated with tectonic plate boundaries.
2. Volcanogenic Massive Sulfide (VMS) Deposits:
- Contrast: VMS deposits form in association with submarine volcanic activity and are characterized by massive sulfide accumulations on the seafloor. CRDs, on the other hand, are often associated with sedimentary environments and result from the replacement of carbonate rocks by ore minerals.
- Commonality: Both VMS and CRDs can contain a variety of base metals, including zinc and lead, and may share some geochemical characteristics.
3. Skarn Deposits:
- Contrast: Skarn deposits form through the interaction of hydrothermal fluids with carbonate rocks, similar to CRDs. However, skarns are typically associated with the intrusion of magmatic rocks, leading to metamorphic changes in the surrounding rocks. CRDs, in contrast, may not have a direct association with intrusive magmatism.
- Commonality: Both deposit types can contain base metals such as zinc, lead, and copper, and may have overlapping mineral assemblages.
4. Sedimentary Exhalative (SEDEX) Deposits:
- Contrast: SEDEX deposits form in sedimentary basins through the exhalation of metal-rich fluids from the seafloor. CRDs, while also associated with sedimentary environments, often involve the replacement of carbonate rocks by ore minerals due to hydrothermal fluids.
- Commonality: Both deposit types can be stratiform and host base metal mineralization, but the specific geological processes leading to their formation differ.
5. Epithermal Gold Deposits:
- Contrast: Epithermal gold deposits form from low-temperature hydrothermal fluids near the Earth’s surface and are characterized by the deposition of gold and silver. CRDs, while involving hydrothermal fluids, are focused on the replacement of carbonate rocks by base metal sulfides.
- Commonality: Both deposit types are associated with hydrothermal processes, and some CRDs may also contain gold and silver as by-products.
6. Stratiform Lead-Zinc Deposits:
- Contrast: Stratiform lead-zinc deposits, similar to SEDEX deposits, are bedded deposits in sedimentary rocks. CRDs, while also occurring in carbonate sequences, may involve more complex hydrothermal replacement processes.
- Commonality: Both deposit types can be stratiform and contain lead and zinc mineralization, but the geological processes leading to their formation can differ.
While these deposit types share some common elements, the distinctions lie in their geological settings, mineralogy, and the specific processes that lead to their formation. Understanding these differences is crucial for effective mineral exploration and resource assessment.
Reference Lists
Books:
- Guilbert, J. M., & Park, C. F. (1986). The Geology of Ore Deposits. Freeman.
- Spry, P. G. (2003). Sulfide Mineralogy and Geochemistry. Cambridge University Press.
- Kesler, S. E., & Wilkinson, B. H. (2008). Earth’s Early Atmosphere and Oceans, and The Origin of Life. Springer.
- Evans, A. M. (1993). Ore Geology and Industrial Minerals: An Introduction. Blackwell Science.
Journal Articles:
- Large, R. R., & Bull, S. W. (2006). Carbonate-hosted lead-zinc deposits. Society of Economic Geologists Special Publication, 10, 307-328.
- Lydon, J. W. (1984). The role of carbonate rocks in the development of Mississippi Valley-type deposits. Economic Geology, 79(3), 321-337.
- Hofstra, A. H. (1995). Skarn deposits. Reviews in Economic Geology, 7, 13-29.
- Hannington, M. D., & Barrie, C. T. (1999). The giant Kidd Creek volcanogenic massive sulfide deposit, western Abitibi subprovince, Canada: a review. Ore Geology Reviews, 14(1), 101-138.
Online Resources:
- Society of Economic Geologists (SEG): https://www.segweb.org/
- Geological Society of America (GSA): https://www.geosociety.org/
- U.S. Geological Survey (USGS): https://www.usgs.gov/
- Australian Mines Atlas – Geoscience Australia: http://www.australianminesatlas.gov.au/