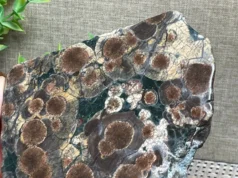
Malachite is a carbonate mineral with chemical composition of Cu2CO3(OH)2. Possibly the earliest ore of copper, malachite is believed to have been mined in the Sinai and eastern deserts of ancient Egypt from as early as 3000 BCE. Single crystals are uncommon; when found, they are short to long prisms. Malachite is usually found as botryoidal or encrusting masses, often with a radiating fibrous structure and banded in various shades of green. It also occurs as delicate fibrous aggregates and as concentrically banded stalactites. Malachite occurs in the altered zones of copper deposits, where it is usually accompanied by lesser amounts of azurite. It is primarily valued as an ornamental material and gemstone. Single masses that weighed up to 51 tons were found in the Ural Mountains of Russia in the 19th century



Name: Derived from the Greek word for mallows, in allusion to its green color.
Crystallography: Monoclinic; prismatic. Crystals usually slender prismatic but seldom distinct. Crystals may be pseudomorphous after azurite. Usually in radiating fibers forming botryoidal or stalactitic masses. Often granular or earthy.
Composition: Basic carbonate of copper, Cu2C03(0H)2. CuO = 71.9 percent, C02 = 19.9 percent, H20 = 8.2 percent. Cu = 57.4 percent
Diagnostic Features: Recognized by its bright green color and botryoidal forms, and distinguished from other green copper minerals by its effervescence in acid
Chemical Properties of Malachite

Malachite is a copper carbonate mineral that has the chemical formula Cu2CO3(OH)2. It is known for its distinctive green color and has a Mohs hardness of 3.5-4. Here are some of the chemical properties of malachite:
- Solubility: Malachite is insoluble in water and most organic solvents. However, it can dissolve in acids such as hydrochloric acid, producing copper chloride, carbon dioxide, and water.
- Stability: Malachite is relatively stable under normal conditions. However, it can decompose at high temperatures to form copper oxide and carbon dioxide.
- Reactivity: Malachite is reactive with acids, such as hydrochloric acid, producing carbon dioxide and copper chloride. It is also reactive with ammonia, forming a deep blue color.
- Conductivity: Malachite is a good conductor of electricity due to its high copper content.
- Oxidation: Malachite is susceptible to oxidation, which can cause its green color to fade over time.
Overall, malachite is a relatively stable mineral with some reactivity towards acids and ammonia. Its conductivity and susceptibility to oxidation are also important chemical properties.
Physical Properties of Malachite
| Color | Bright green, dark green, blackish green, commonly banded in masses; green to yellowish green in transmitted light |
| Streak | Light green |
| Luster | Adamantine to vitreous; silky if fibrous; dull to earthy if massive |
| Cleavage | Perfect on {201}, fair on {010}. |
| Diaphaneity | Translucent to opaque |
| Mohs Hardness | 3.5–4.0 |
| Specific Gravity | 3.6–4 |
| Diagnostic Properties | Green color, soft, effervesces with dilute HCl to produce a green liquid. |
| Crystal System | Monoclinic |
| Tenacity | Brittle |
| Fracture | Irregular/Uneven, Sub-Conchoidal, Fibrous |
| Density | 3.6 – 4.05 g/cm3 (Measured) 4 g/cm3 (Calculated) |
Optical Properties of Malachite
| Type | Anisotropic |
| Color / Pleochroism | Visible |
| Twinning | Common as contact or penetration twins on {100} and {201}. Polysynthetic twinning also present. |
| Optic Sign | Biaxial (-) |
| Birefringence | δ = 0.254 |
| Relief | Very High |
Occurrence and Formation

Malachite is a popular green mineral known for its distinctive color and unique banded patterns. It is primarily composed of copper carbonate hydroxide [Cu2CO3(OH)2]. Malachite forms under specific geological conditions and is often associated with copper deposits. Here’s an overview of its occurrence and formation:
- Geological Setting: Malachite typically occurs in copper-rich environments, especially in regions where copper minerals are concentrated. It is often found alongside other copper minerals like azurite, chrysocolla, and cuprite.
- Primary Formation: Malachite forms through the weathering and oxidation of primary copper minerals, such as chalcopyrite (copper iron sulfide) and bornite (copper iron sulfide). These primary minerals are exposed to oxygen, carbon dioxide, and water, leading to chemical reactions that convert them into secondary copper minerals, including malachite.
- Chemical Reactions: The formation of malachite involves several chemical reactions. Initially, the primary copper minerals react with oxygen and water to form copper ions (Cu2+). These copper ions then combine with carbonate ions (CO3^2-) from sources like groundwater or rainwater to create copper carbonate compounds, including malachite.The reactions can be summarized as follows:
- CuFeS2 (chalcopyrite) + O2 + H2O → Cu2+ + 2Fe2+ + 2SO4^2- + 2H+
- Cu2+ + CO3^2- → CuCO3 (copper carbonate)
- Hydrothermal Activity: Malachite can also form in hydrothermal environments where hot, mineral-rich fluids flow through fractures in rocks. In these settings, copper minerals dissolved in the hydrothermal fluids can precipitate out and form malachite deposits.
- Secondary Alteration: Malachite is often associated with secondary alteration zones near the surface, where copper minerals in rocks have been leached, oxidized, and transformed into secondary copper minerals. These alteration zones can be found in various geological settings, such as sedimentary rocks, igneous rocks, and hydrothermal veins.
- Vein Deposits: In some cases, malachite can be found in veins or fractures within rocks, where it forms as a result of the interaction between copper-rich fluids and host rocks.
- Associations: Malachite can be associated with other secondary copper minerals like azurite (another copper carbonate mineral) and chrysocolla (a hydrous copper silicate), which often share similar formation conditions.
It’s important to note that malachite can also be found as a secondary mineral in oxidized copper ore deposits, often occurring as crusts, coatings, or botryoidal (grape-like) masses on the surface of rocks. Its beautiful green color and unique patterns make it a popular mineral for lapidary purposes, jewelry, and ornamental objects.
Malachite Application and Uses

Malachite has been used for various purposes throughout history due to its attractive green color and unique banded patterns. Its applications and uses include:
- Ornamental and Decorative Use: Malachite is highly prized as a gemstone and ornamental material. It is often carved into beads, cabochons, figurines, and decorative items. Its intricate green patterns make it a popular choice for jewelry, such as necklaces, pendants, rings, and earrings.
- Inlay and Mosaic Work: Malachite’s vibrant green color and swirling patterns make it an excellent choice for inlay work and mosaics in architectural and artistic applications. It has been used to decorate furniture, walls, and architectural details.
- Healing and Metaphysical Properties: Some people believe that malachite possesses healing and metaphysical properties. It is associated with protection, emotional balance, and spiritual growth. Malachite is often used in crystal healing and as a talisman or amulet.
- Pigments: Historically, malachite has been ground into a fine powder to create green pigments for painting and dyeing. The pigment was used in ancient civilizations for artistic and decorative purposes.
- Collectibles: Collectors often seek out high-quality malachite specimens and carvings due to their beauty and rarity. Unique patterns and large specimens can be valuable collectibles.
- Historical and Cultural Significance: Malachite has played a role in various cultures throughout history. It has been used in the creation of religious artifacts, jewelry, and decorative objects in ancient civilizations such as Egypt, Greece, and Rome.
- Lapidary Art: Malachite is a popular choice among lapidary artists who shape and polish stones to create intricate and unique designs. It is used in lapidary arts to make gemstones and cabochons.
- Mineral Specimen: Malachite is highly regarded as a mineral specimen for educational and display purposes. Museums and collectors often showcase malachite specimens to illustrate mineralogy and geology.
- Scientific Research: Malachite is of interest to geologists and mineralogists for its crystal structure and formation. Its study can provide insights into geological processes, especially those related to the weathering and alteration of copper minerals.
- Metallurgy: In some cases, malachite can be a source of copper ore. Historically, it has been used as an ore for copper extraction, although it is not a primary source due to the relatively low copper content.
It’s important to note that while malachite has been historically used for some of these purposes, its use in modern pigments, for example, has largely been replaced by synthetic alternatives due to environmental and toxicity concerns. Additionally, when handling malachite, it’s important to be aware that it contains copper, which can be toxic if ingested or inhaled, so proper precautions should be taken.
Malachite Notable Deposits

Malachite is found in various locations around the world, often associated with copper deposits and secondary copper minerals. Here are some notable deposits and regions where malachite is commonly found:
- Democratic Republic of the Congo (DRC): The DRC, particularly the Katanga Province, is known for its rich copper deposits, and malachite is frequently found alongside other copper minerals like azurite and cuprite.
- Australia: Malachite deposits are found in several Australian states, including Queensland, New South Wales, South Australia, and Western Australia. Prominent locations include the Mount Isa region in Queensland and the Broken Hill area in New South Wales.
- Russia: Malachite is found in various regions of Russia, with notable deposits in the Ural Mountains and the Siberian region. The Ural Mountains are particularly famous for malachite mining.
- United States: Malachite can be found in various states across the U.S., including Arizona, New Mexico, Nevada, and Utah. The southwestern United States is known for its copper deposits and associated copper minerals, including malachite.
- Namibia: Malachite deposits are found in the Tsumeb Mine, which is renowned for its diverse mineral specimens. The mine has produced exceptionally well-preserved malachite specimens.
- Zambia: Zambia is a significant copper-producing country in Africa, and malachite can be found in copper mines and associated deposits.
- Chile: Malachite is associated with copper deposits in Chile, which is one of the world’s largest copper producers.
- China: China has malachite deposits in various regions, including Yunnan and Guangdong provinces. Chinese malachite is often used for carving and ornamental purposes.
- Morocco: Malachite is found in Morocco, especially in the Atlas Mountains and the Tazalarht region.
- Mexico: Mexican malachite is known for its vivid green color and is found in various locations, including Sonora, Chihuahua, and Durango.
- Kazakhstan: Malachite can be found in some copper mining areas in Kazakhstan.
- Australia: Malachite deposits are found in several Australian states, including Queensland, New South Wales, South Australia, and Western Australia. Prominent locations include the Mount Isa region in Queensland and the Broken Hill area in New South Wales.
- United States: Malachite can be found in various states across the U.S., including Arizona, New Mexico, Nevada, and Utah. The southwestern United States is known for its copper deposits and associated copper minerals, including malachite.
These are just a few notable locations where malachite can be found, but it can also be encountered in other parts of the world where copper-rich environments exist. Mineral collectors and enthusiasts often seek out malachite specimens from these regions due to their beauty and unique patterns.
References
- Bonewitz, R. (2012). Rocks and minerals. 2nd ed. London: DK Publishing.
- Handbookofmineralogy.org. (2019). Handbook of Mineralogy. [online] Available at: http://www.handbookofmineralogy.org [Accessed 4 Mar. 2019].
- Mindat.org. (2019). Malachite: Mineral information, data and localities.. [online] Available at: https://www.mindat.org/min-727.html [Accessed 4 Mar. 2019].