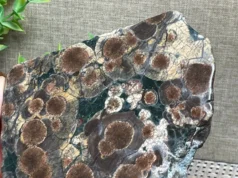
Pyrite is commonly referred to as “fool’s gold.” Although much lighter than gold, its brassy color and relatively high density misled many novice prospectors. Its name is derived from the Greek word pyr, meaning “fire,” because it emits sparks when struck by iron. It is opaque and pale silvery yellow when fresh, turning darker and tarnishing with exposure to oxygen. Pyrite crystals may be cubic, octahedral, or twelve-sided “pyritohedra,” and are often striated. Pyrite can also be massive or granular, or form either flattened disks or nodules of radiating, elongate crystals. Pyrite occurs in hydrothermal veins, by segregation from magmas, in contact metamorphic rocks, and in sedimentary rocks, such as shale and coal, where it can either fill or replace fossils.



Name: From the Greek for fire, as sparks may be struck from it.
Polymorphism & Series: Dimorphous with marcasite; forms a series with cattierite.
Mineral Group: Pyrite group.
Association: Pyrrhotite, marcasite, galena, sphalerite, arsenopyrite, chalcopyrite, many other sulfides and sulfosalts, hematite, fluorite, quartz, barite, calcite.
Pyrite formation and occurrence

Pyrite, also known as “fool’s gold,” is a common iron sulfide mineral with the chemical formula FeS2. It forms in a variety of geological settings through several processes.
Formation: Pyrite forms under reducing conditions, typically in environments with low oxygen levels, high sulfur content, and abundant iron. It can form through both biological and abiotic processes.
- Hydrothermal processes: Pyrite can form from hydrothermal fluids, which are hot, mineral-rich fluids that circulate through fractures in rocks. As these fluids cool and react with the surrounding rock, pyrite can precipitate from the solution.
- Sedimentary processes: Pyrite can also form in sedimentary environments, such as in marine sediments or coal deposits. Organic matter in sediments can release sulfur as it decays, which can react with iron minerals to form pyrite.
- Metamorphic processes: Pyrite can form during metamorphism, which is the process of changes in mineralogical, chemical, and textural characteristics of rocks due to high temperature and pressure. Pyrite can form during regional or contact metamorphism, where existing iron-rich minerals are subjected to heat and pressure, leading to the formation of pyrite.
Occurrence: Pyrite occurs worldwide in a wide range of geological settings, including:
- Igneous rocks: Pyrite can be found in igneous rocks, such as granite, gabbro, and basalt, particularly in hydrothermal veins and disseminated throughout the rock.
- Sedimentary rocks: Pyrite is commonly found in sedimentary rocks, such as shale, sandstone, and limestone, as nodules, concretions, or disseminated grains.
- Metamorphic rocks: Pyrite can be present in metamorphic rocks, such as schist, gneiss, and slate, formed through the metamorphism of existing sedimentary rocks or other pyrite-containing minerals.
- Hydrothermal veins: Pyrite can occur in hydrothermal veins associated with various types of ore deposits, including gold, copper, and lead-zinc deposits.
- Coal deposits: Pyrite is often associated with coal deposits, where it forms as a result of organic matter decomposition and subsequent mineralization.
It’s important to note that pyrite can be unstable in certain environments and can oxidize, forming sulfuric acid and iron oxide minerals, which can lead to environmental issues such as acid mine drainage.
Chemical Properties of Pyrite
Pyrite, with the chemical formula FeS2, is a mineral that possesses several chemical properties. Some of the key chemical properties of pyrite are:
- Composition: Pyrite is composed of iron (Fe) and sulfur (S) atoms in a 1:2 ratio, with two sulfur atoms for every one iron atom. Its chemical formula is FeS2, indicating that it consists of an iron atom bonded to two sulfur atoms.
- Crystal structure: Pyrite crystallizes in the cubic system and belongs to the isometric crystal class. It has a distinctive cubic or octahedral crystal shape, with a brassy-yellow to pale-gold color and a metallic luster.
- Hardness: Pyrite has a hardness of 6 to 6.5 on the Mohs scale, which means it is relatively hard and can scratch glass.
- Density: The density of pyrite is around 4.8 to 5.0 g/cm^3, which is relatively heavy compared to many other minerals.
- Magnetism: Pyrite is weakly magnetic, meaning it can be attracted to a magnet. However, its magnetic properties are typically not very strong.
- Reactivity: Pyrite is relatively inert and does not easily react with water or most acids. However, it can slowly oxidize in the presence of oxygen and water, forming sulfuric acid and iron oxide minerals, which can result in the formation of acid mine drainage in certain environments.
- Thermal properties: Pyrite has a relatively high melting point of around 1,070°C (1,958°F), which means it can withstand high temperatures without undergoing significant changes in its chemical composition.
- Pyrophoricity: Pyrite can exhibit pyrophoric properties, meaning it can ignite spontaneously in the presence of air or oxygen. This can make pyrite a fire hazard in certain conditions.
- Isomorphism: Pyrite can exhibit isomorphism, which is the ability to form solid solutions with other minerals, such as marcasite (FeS2), which is a polymorph of pyrite with a different crystal structure but similar chemical composition.
These are some of the main chemical properties of pyrite, which contribute to its unique characteristics and behavior in various geological and environmental settings.
Physical Properties of Pyrite
| Color | Pale brass-yellow reflective; tarnishes darker and iridescent |
| Streak | Greenish-black to brownish-black |
| Luster | Metallic, glistening |
| Cleavage | Poor/Indistinct Indistinct on {001}. |
| Diaphaneity | Opaque |
| Mohs Hardness | 6–6.5 |
| Specific Gravity | 4.95–5.10 |
| Crystal System | Isometric |
| Tenacity | Brittle |
| Fracture | Irregular/Uneven, Conchoidal |
| Density | 4.8 – 5 g/cm3 (Measured) 5.01 g/cm3 (Calculated) |
Optical Properties of Pyrite

- Color: Pyrite typically has a brassy-yellow to pale-gold color, although it can also appear silver, bronze, or even black in some cases, depending on impurities and weathering. Its color is often one of the most distinctive features of pyrite.
- Luster: Pyrite has a metallic luster, meaning it reflects light in a manner similar to a metal, giving it a shiny appearance.
- Transparency: Pyrite is generally opaque, meaning it does not transmit light and is not transparent.
- Diaphaneity: Pyrite is typically diaphanous, which means it does not allow light to pass through it.
- Birefringence: Pyrite is not birefringent, which means it does not exhibit double refraction of light when viewed under a polarizing microscope.
- Pleochroism: Pyrite is usually not pleochroic, meaning it does not display different colors when viewed from different angles under plane-polarized light.
- Refractive index: The refractive index of pyrite is relatively high, typically ranging from around 2.5 to 2.7, depending on the wavelength of light and the crystal orientation.
- Dispersion: Pyrite has a relatively low dispersion, which means it does not significantly separate light into its component colors when viewed under a dispersive prism or in a spectroscope.
- Fluorescence: Pyrite does not typically exhibit fluorescence under ultraviolet (UV) light.
These are some of the main optical properties of pyrite, which can be used to identify and characterize this mineral in various geological and mineralogical contexts. It’s important to note that optical properties of pyrite can vary depending on factors such as crystal size, impurities, and weathering, and careful examination using appropriate optical techniques and equipment is necessary for accurate identification.
Pyrite Uses

Pyrite has been used for various purposes throughout history due to its unique properties. Some of the main uses of pyrite include:
- Jewelry and Decorative Items: Pyrite’s brassy-yellow to pale-gold color and metallic luster make it a popular material for jewelry and decorative items. It has been used to create gemstones, beads, cabochons, and other ornamental pieces. Pyrite has also been used in jewelry designs as a substitute for gold due to its resemblance to gold, hence the nickname “fool’s gold.”
- Sulfur Production: Pyrite is a major source of sulfur, which is an important element used in the production of various chemicals, such as sulfuric acid, fertilizers, and detergents. Pyrite can be burned to produce sulfur dioxide gas, which can then be converted into sulfuric acid through chemical processes.
- Industrial Applications: Pyrite has been used in various industrial applications, such as in the production of iron and steel. Pyrite can be used as a source of iron in the production of iron ore pellets, which are used as raw materials in the manufacturing of steel. Pyrite has also been used as a component in the production of sulfur-containing chemicals, as a catalyst in certain chemical reactions, and as a material for producing sparklers and fireworks due to its ability to create sparks when struck against a hard surface.
- Geological and Mineralogical Studies: Pyrite is a common mineral in many geological formations and is often used as an indicator mineral in geological and mineralogical studies. Its presence or absence, as well as its characteristics, can provide valuable information about the geological history, mineralization processes, and hydrothermal activities of a particular area.
It’s important to note that while pyrite has various uses, it is not always suitable for all applications. Pyrite can oxidize and release sulfuric acid, leading to potential environmental issues such as acid mine drainage when exposed to air and water. Therefore, proper care and consideration of environmental impacts should be taken into account when using pyrite for any purpose.
Distribution
Pyrite is a widespread mineral and is found in various geological formations around the world. It occurs in a wide range of environments and can be found in both sedimentary and igneous rocks, as well as in hydrothermal veins and metamorphic rocks. Some of the main distribution areas of pyrite include:
- Sedimentary Rocks: Pyrite can be found in sedimentary rocks, such as shale, coal, and limestone, in many parts of the world. In coal deposits, pyrite is commonly present as small nodules or bands known as “pyrite framboids” and can sometimes be responsible for the spontaneous combustion of coal due to its ability to oxidize and generate heat.
- Igneous Rocks: Pyrite can also be found in some igneous rocks, particularly those that are rich in iron and sulfur. It can occur in various types of igneous rocks, such as granite, diorite, gabbro, and basalt, and is often associated with other sulfide minerals.
- Hydrothermal Veins: Pyrite is a common mineral in hydrothermal veins, which are formed when hot fluids containing dissolved minerals migrate through fractures in rocks and precipitate minerals as they cool. Pyrite can be found in hydrothermal veins associated with ore deposits of various types, such as copper, gold, zinc, lead, and silver.
- Metamorphic Rocks: Pyrite can also form in metamorphic rocks, which are formed when existing rocks are subjected to high temperature and pressure conditions. Pyrite can occur in different types of metamorphic rocks, such as schist, gneiss, and slate, and is often associated with other sulfide minerals.
- Geological Formations: Pyrite can be found in various geological formations, such as concretions, nodules, and concretional nodules, in different parts of the world. These formations can occur in sedimentary rocks, soils, and other environments, and can have distinctive shapes and sizes.
Pyrite is a widely distributed mineral, and its occurrence can vary depending on local geology, mineralization processes, and geological history. It is important to note that the distribution of pyrite can also be influenced by factors such as weathering, erosion, and human activities, and proper exploration and sampling techniques should be employed for accurate identification and characterization of pyrite occurrences in specific locations
References
- Bonewitz, R. (2012). Rocks and minerals. 2nd ed. London: DK Publishing.
- Handbookofmineralogy.org. (2019). Handbook of Mineralogy. [online] Available at: http://www.handbookofmineralogy.org [Accessed 4 Mar. 2019].
- Mindat.org. (2019). Pyrite: Mineral information, data and localities.. Available at: https://www.mindat.org/