Hematite is one of the most defining minerals in Earth’s geological and industrial history. Known for its striking metallic luster, its surprising weight, and its unmistakable deep red streak, hematite stands as the most stable and widespread iron oxide on the planet. It appears in volcanic terrains, sedimentary basins, hydrothermal veins, metamorphic environments, and even in atmospheric oxidation crusts on the surface of basalt. Few minerals occur in such diversity, and even fewer have had as much impact on both planetary evolution and human civilization.
Beyond its scientific relevance, hematite has been used for tens of thousands of years as pigment, ornament, symbolic material, and today as the main source of iron for steel production. Although its outward appearance varies dramatically—from mirror-like silver plates to earthy red powders—its internal physical and optical properties remain consistent and diagnostic.
This article provides a comprehensive and global overview of hematite, including its formation, mineralogical characteristics, optical behavior, varieties, geological significance, and modern industrial uses.
1. Definition and Mineral Identity

Hematite is an iron oxide mineral with the chemical formula Fe₂O₃. It belongs to the oxide mineral group and crystallizes in the trigonal system. Its name originates from the Greek word haima, meaning “blood,” referring to the red coloration produced when hematite is scratched or powdered.
Although hematite frequently appears metallic-gray or black, it is chemically identical to the red ochre used in prehistoric art. Its streak—always red—remains the single most definitive diagnostic feature.
2. Geological Formation of Hematite

Hematite forms through a wide range of processes, all of which involve the oxidation of iron. Because oxygen is abundant in the atmosphere and hydrosphere, hematite naturally develops in environments spanning deep-sea sediments, continental basins, volcanic terrains, and hydrothermal systems.
2.1. Sedimentary Formation
Sedimentary hematite forms through:
- chemical precipitation of iron from seawater
- oxidation of dissolved iron during diagenesis
- weathering and oxidation of iron-rich minerals
Much of the red color found in sandstones, shales, and ironstones is due to fine-grained hematite coating sediment grains.
2.2. Banded Iron Formations (BIFs)
The world’s largest hematite deposits occur within Precambrian banded iron formations. These ancient layers record a dramatic shift in Earth’s atmospheric composition during the Great Oxygenation Event, when oxygen produced by microbial life reacted with iron in the oceans. The resulting hematite and magnetite precipitated into alternating iron-rich and silica-rich layers, forming deposits now mined globally.
2.3. Hydrothermal Hematite
In hydrothermal systems, hot aqueous solutions dissolve iron from surrounding rocks. As these fluids cool or mix with oxygenated water, iron precipitates as hematite. Hydrothermal hematite often forms metallic, specular, or massive aggregates.
2.4. Metamorphic Hematite
Metamorphic hematite forms through:
- oxidation of magnetite
- recrystallization of sedimentary iron minerals
- high-pressure alteration of iron-rich layers
Metamorphic hematite often displays platy, reflective crystals.
2.5. Surface and Volcanic Hematite
Volcanic rocks such as basalt and andesite weather rapidly when exposed to oxygen. Iron within the rock oxidizes into hematite, forming reddish coatings or alteration rinds on rock surfaces.
3. Varieties of Hematite

Despite having a single chemical composition, hematite exhibits remarkable diversity in appearance.
3.1. Specular Hematite (Specularite)
- Mirror-like metallic luster
- Shiny, reflective plates
- Common in metamorphic and hydrothermal deposits
3.2. Metallic Hematite
- Silver-gray surface
- Strong metallic reflection
- Dense, massive habit
- Commonly polished for jewelry
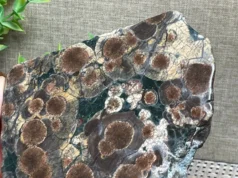
3.3. Botryoidal Hematite
- Rounded, grape-like structures
- Smooth and glossy surfaces
- Often formed in low-temperature aqueous environments
3.4. Oolitic Hematite
- Small spherical grains (oolites) cemented together
- Usually reddish-brown
- Common in sedimentary ironstones
3.5. Earthy Red Hematite (Red Ochre)
- Fine-grained, powdery
- Deep red color
- Used for pigment since prehistoric times
3.6. Martite
- Hematite pseudomorph after magnetite
- Crystal shape preserved but composition altered to Fe₂O₃
4. Physical and Optical Properties of Hematite

Hematite’s internal properties are consistent across all forms, regardless of external color or habit.
4.1. Physical Behavior
Hematite is unusually dense due to its high iron content. It is moderately hard, brittle, and exhibits a range of surface lusters. Although many samples look metallic, others appear dull, earthy, or red.
4.2. Optical Characteristics
Hematite has distinctive optical behavior, including:
- Opaque transparency in nearly all forms
- Strong metallic reflection in specular varieties
- Submetallic to earthy luster in red or massive forms
- Red internal coloration observable when powdered
- High refractive index, giving hematite its characteristic dark, glassy appearance
- Anisotropic optical response in platy or lamellar crystals
- No pleochroism, as hematite is opaque
Under reflected light microscopy, hematite shows:
- brilliant metallic reflectance
- high polish
- distinct red internal reflections in thin edges
These optical traits are crucial in ore petrography for distinguishing hematite from magnetite, goethite, and ilmenite.
5. Physical Properties Table

Below is a globally standardized reference table for hematite’s physical and optical properties.
| Property | Value / Description |
|---|---|
| Chemical Formula | Fe₂O₃ |
| Mineral Class | Oxide |
| Crystal System | Trigonal |
| Color | Silver-gray, black, red, reddish-brown |
| Streak | Red to reddish-brown (diagnostic) |
| Luster | Metallic, semi-metallic, earthy |
| Transparency | Opaque |
| Hardness (Mohs) | 5.5 – 6.5 |
| Density / Specific Gravity | ~5.26 g/cm³ |
| Cleavage | None |
| Fracture | Subconchoidal to uneven |
| Magnetic Behavior | Nonmagnetic (pure hematite) |
| Optical Reflectivity | Strong metallic in specular varieties |
| Refractive Index (n) | Very high; variable due to opacity |
| Common Habits | Botryoidal, tabular, massive, specular |
| Internal Color | Deep red in powdered form |
| Iron Content | Up to 70% Fe |
6. Geological and Planetary Significance
Hematite is a key indicator mineral in geological studies. Because it forms under oxidizing conditions, its presence in ancient rocks marks the evolution of Earth’s atmosphere and hydrosphere.
In planetary science, hematite is also notable because Mars has abundant hematite deposits, contributing to its red coloration. NASA rover analyses have confirmed both fine-grained red hematite and crystalline specular hematite on the Martian surface.
7. Economic Importance: The Foundation of Global Iron Production
Hematite is the world’s primary iron ore. High-grade ores often contain between 60% and 68% Fe, making them highly efficient for steelmaking. Major global mining regions include:
- Western Australia (Pilbara)
- Brazil (Carajás)
- South Africa
- India
- Canada
- Russia
The global steel industry — construction, transportation, manufacturing, energy infrastructure — depends heavily on hematite deposits formed billions of years ago.
8. Hematite in Jewelry and Design
Polished hematite is widely used in jewelry due to its:
- mirror-like metallic finish
- high density and weight
- smooth polish
- sleek modern appearance
However, many products marketed as “hematite” or “magnetic hematite” are actually synthetic ferrite ceramics, not natural Fe₂O₃.
9. Cultural and Symbolic Uses
Hematite has been used for symbolic and artistic purposes for tens of thousands of years. Red ochre from hematite was used in cave paintings, rituals, burials, and early cosmetics. Today it remains popular in metaphysical communities as a “grounding” stone, though such claims are not scientifically supported.
10. Identification Guide
Hematite is easy to identify using simple field tests:
- Streak: Always red
- Density: Very heavy for its size
- Magnetism: Not magnetic
- Appearance: Metallic or earthy, depending on variety
Specular hematite will reflect light sharply, while earthy forms appear dull.
Conclusion
Hematite is a mineral that bridges planetary evolution, human history, and modern industry. Its formation records the oxygenation of ancient oceans. Its durability and abundance fuel global steel production. Its pigment has colored human culture for tens of thousands of years. And its physical and optical properties continue to make it one of the most studied and recognized minerals in geology.
Whether found as shimmering metallic plates or as red ochre dust, hematite remains one of the most important minerals on Earth.