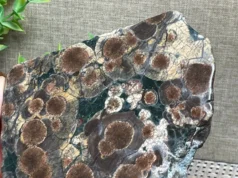
Smithsonite is a mineral that belongs to the carbonate group. It is named after James Smithson, an English scientist and mineralogist who was the founder of the Smithsonian Institution in the United States. Smithsonite is also known by several other names, including zinc spar, zinc carbonate, and calamine.



The mineral is composed of zinc carbonate (ZnCO3) and usually occurs in a variety of colors, including shades of blue, green, pink, gray, and white. Its colors can be vibrant and attractive, making it a popular choice for gemstone collectors and jewelry enthusiasts.
Smithsonite is typically found in the oxidized zone of zinc ore deposits, where it forms as a secondary mineral. It often occurs as botryoidal (grape-like) or stalactitic formations, with a characteristic rounded or bulbous shape. The mineral can also be found in crusts, veins, and other forms.
One of the distinctive properties of smithsonite is its effervescence when it comes into contact with dilute hydrochloric acid. This reaction is due to the presence of carbonate ions in its chemical composition.
Historically, smithsonite has been mined as an ore of zinc, which is an important industrial metal used in various applications, including galvanizing steel, producing batteries, and making alloys. However, the mineral’s aesthetic appeal and vibrant colors have also made it a sought-after material in the world of gemstones and mineral collecting.
Smithsonite can be cut and polished to create attractive cabochons, beads, and other gemstone forms. It is relatively soft, with a Mohs hardness of 4 to 5, which means it can be scratched by harder materials. Therefore, it requires proper care to avoid damage.
Due to its association with zinc, smithsonite has also been used in traditional medicine as a remedy for various ailments. In particular, it has been employed for its alleged healing properties in treating skin conditions, such as eczema and rashes.
In summary, smithsonite is a carbonate mineral that is prized for its vibrant colors, making it a popular choice for gemstone collectors and jewelry makers. It is named after James Smithson, the founder of the Smithsonian Institution, and has a historical significance as an ore of zinc. Whether appreciated for its aesthetics or its industrial uses, smithsonite continues to captivate people with its unique beauty and properties.
Historical background
Smithsonite has an interesting historical background closely tied to its namesake, James Smithson. James Smithson was an English chemist and mineralogist born in 1765. He conducted extensive research in various scientific fields, including mineralogy and chemistry.
Smithson is best known for his bequest to the United States, which eventually led to the establishment of the Smithsonian Institution. In his will, Smithson left his entire estate to his nephew, with the stipulation that if his nephew were to die without any heirs, the estate should go “to the United States of America, to found at Washington, under the name of the Smithsonian Institution, an establishment for the increase and diffusion of knowledge among men.”
Smithson’s bequest was unexpected, as he had no known connection to the United States during his lifetime. The reason behind his decision remains a subject of speculation. Some theories suggest that Smithson’s interest in America may have been influenced by his interactions with prominent American scientists and intellectuals of his time.
After Smithson’s death in 1829, his nephew died childless, and as a result, his estate, including his vast collection of minerals, passed to the United States. The bequest created a significant dilemma for American lawmakers, as they had to determine how best to fulfill Smithson’s wishes and establish an institution for the “increase and diffusion of knowledge.”
In 1846, the United States Congress passed an act to officially accept the bequest and establish the Smithsonian Institution. The Institution’s initial focus was on scientific research, exploration, and education. Today, the Smithsonian Institution is a renowned complex of museums, research centers, and educational programs dedicated to preserving and disseminating knowledge across a wide range of disciplines.
James Smithson’s contributions to mineralogy and his collection of minerals, including the mineral that would later be named smithsonite in his honor, played a role in the development of the Smithsonian Institution. Although smithsonite was discovered prior to the establishment of the Institution, its naming serves as a lasting tribute to Smithson’s passion for scientific exploration and his enduring legacy in the field of knowledge dissemination.
Significance and uses
- Gemstone and Jewelry: Smithsonite’s vibrant colors, ranging from blues and greens to pinks and grays, make it highly valued in the world of gemstones and jewelry. It is often cut and polished to create attractive cabochons, beads, and other gemstone forms. Its beauty and uniqueness make it a sought-after choice for collectors and jewelry enthusiasts.
- Mineral Collecting: Smithsonite is highly prized by mineral collectors due to its diverse colors, crystal habits, and associations with other minerals. Specimens with well-formed crystals or interesting formations are highly sought after and can be valuable additions to mineral collections.
- Industrial Uses: As a mineral composed of zinc carbonate, smithsonite has historically been mined as an ore of zinc. Zinc is an important industrial metal used in various applications. Smithsonite has been a source of zinc for the production of metallic zinc, as well as for other zinc compounds. It is used in galvanizing steel, producing batteries, making alloys, and other zinc-related industries.
- Scientific Research: Smithsonite, like other minerals, has been the subject of scientific research. Its crystal structure, formation processes, and properties have been studied to enhance our understanding of mineralogy, crystallography, and geochemistry. Research on smithsonite contributes to our knowledge of Earth’s processes and the formation of mineral deposits.
- Historical and Cultural Significance: Smithsonite holds historical significance due to its association with James Smithson, the founder of the Smithsonian Institution. The naming of the mineral after him and its connection to the establishment of the Institution adds a layer of historical and cultural importance to smithsonite.
In summary, smithsonite’s significance lies in its beauty as a gemstone, its value in mineral collections, its industrial uses as a source of zinc, its traditional medicinal applications, its contribution to scientific research, and its historical association with the Smithsonian Institution.
Physical Properties of Smithsonite
- Color: Smithsonite occurs in a variety of colors, including shades of blue, green, pink, gray, and white. The colors can be vibrant and may exhibit variations within a single specimen.
- Crystal System: Smithsonite crystallizes in the trigonal crystal system. However, it is more commonly found in botryoidal (grape-like) or stalactitic formations rather than as distinct crystals.
- Hardness: Smithsonite has a hardness of 4 to 5 on the Mohs scale. This means it is relatively soft and can be scratched by harder materials.
- Cleavage: Smithsonite exhibits a perfect rhombohedral cleavage. This means that it can be easily cleaved into rhombohedral fragments with smooth, flat surfaces.
- Fracture: The mineral displays a conchoidal or uneven fracture, meaning it breaks with curved, shell-like surfaces or irregularly.
- Luster: Smithsonite has a vitreous to pearly luster. The luster refers to the way light reflects off the surface of the mineral.
- Transparency: Smithsonite is typically translucent to opaque, with varying degrees of transparency depending on the specific specimen.
- Specific Gravity: The specific gravity of smithsonite ranges from approximately 4 to 4.4. This value indicates that it is relatively dense compared to water.
- Streak: The streak of smithsonite is usually white, regardless of the color of the mineral itself. The streak is the color left behind when the mineral is scratched on an unglazed porcelain plate.
- Effervescence: One notable characteristic of smithsonite is its effervescence when it comes into contact with dilute hydrochloric acid. The mineral produces bubbles due to the release of carbon dioxide gas resulting from the reaction with the acid. This effervescence confirms the presence of carbonate ions in its chemical composition.
These physical properties collectively contribute to the identification and characterization of smithsonite in both scientific and practical contexts.
Occurrence and Formation of Smithsonite
Smithsonite occurs in various geological settings and is primarily formed as a secondary mineral through the alteration of primary zinc minerals. Here is information about the occurrence and formation of smithsonite:
- Geological Settings: Smithsonite is commonly found in oxidized zinc ore deposits. It occurs in association with other zinc minerals such as sphalerite, hemimorphite, and willemite. It can also be found in lead-zinc deposits, as well as in the oxidized zones of zinc-rich hydrothermal veins and replacement deposits.
- Formation Process: Smithsonite forms through the chemical alteration of primary zinc minerals, especially sphalerite (zinc sulfide). When primary zinc minerals are exposed to weathering and groundwater rich in carbonates, chemical reactions occur, leading to the breakdown of sphalerite and the precipitation of smithsonite.
- Carbonation Process: The formation of smithsonite involves a process called carbonation, where carbon dioxide (CO2) from the atmosphere or from dissolved carbon dioxide in water reacts with primary zinc minerals in the presence of water. This reaction results in the replacement of zinc sulfide or other primary zinc minerals with zinc carbonate, forming smithsonite.
- Environment: Smithsonite commonly forms in environments with oxidizing conditions and the presence of carbonates. This includes weathered and oxidized zones near the surface, such as limestone caves, mine tailings, and areas affected by hydrothermal alteration.
- Crystal Habits: Smithsonite can exhibit various crystal habits, although it is more commonly found in massive, botryoidal (grape-like), stalactitic, or crust-like forms. These formations arise from the precipitation of smithsonite within cavities, fractures, or voids in the host rock.
- Colors: The colors of smithsonite can vary due to impurities present during its formation. Different metal ions or trace elements can impart shades of blue, green, pink, gray, and white to the mineral.
- Replacement Deposits: Smithsonite can also occur as a replacement mineral within host rocks, replacing pre-existing minerals. This process can lead to the preservation of the original shape or texture of the replaced mineral.
It’s worth noting that the occurrence and formation of smithsonite can vary depending on the specific geological conditions of each deposit. Understanding these processes is crucial for both scientific research and mineral exploration, as it helps in identifying potential ore deposits and assessing the economic viability of mining operations.
Varieties and Colors of Smithsonite
Smithsonite exhibits a wide range of colors and variations, making it a visually appealing mineral. The color of smithsonite is primarily influenced by impurities and trace elements present during its formation. Here are some of the notable varieties and colors of smithsonite:
- Blue Smithsonite: Blue is one of the most well-known and sought-after colors of smithsonite. It can range from pale blue to deep azure or even turquoise. The blue color is often caused by the presence of copper impurities within the crystal structure.
- Green Smithsonite: Green smithsonite is another popular variety, which can range from light mint green to rich emerald green. The green color is typically associated with the presence of iron or copper impurities.
- Pink Smithsonite: Pink smithsonite is valued for its delicate and soft pink hues. The pink color is often caused by the presence of manganese or cobalt impurities.
- Gray and White Smithsonite: Smithsonite can also occur in shades of gray and white. These lighter colors often result from the absence of significant impurities or the presence of minor amounts of other elements.
- Multicolored Smithsonite: Smithsonite can display multicolored patterns and banding, with combinations of blue, green, pink, and white occurring within a single specimen. These multicolored varieties are particularly prized by collectors for their unique and attractive appearances.
It’s important to note that the exact color and variations of smithsonite can differ depending on the specific locality where it is found. Different geological conditions and the presence of specific impurities contribute to the diverse range of colors observed in smithsonite specimens.
The vibrant and diverse color palette of smithsonite, combined with its attractive crystal habits, makes it a highly sought-after mineral for gemstone enthusiasts, collectors, and jewelry makers. The unique colors and patterns of smithsonite specimens make each piece truly unique and visually captivating.
Industrial and Commercial Uses of Smithsonite
Smithsonite, as a mineral composed of zinc carbonate, has several industrial and commercial uses. Here are some of the key applications:
- Zinc Production: Smithsonite has been historically mined as an ore of zinc, an important industrial metal. Zinc is primarily used for galvanizing steel, a process that protects steel from corrosion by coating it with a layer of zinc. This helps extend the lifespan of steel structures, such as buildings, bridges, and pipelines.
- Alloy Manufacturing: Zinc is also used in the production of various alloys. Smithsonite can be a source of zinc for alloy manufacturing, where it is combined with other metals to enhance their properties. For example, zinc is commonly used in brass, a copper-zinc alloy, to improve corrosion resistance and create a golden appearance.
- Chemical Industry: Smithsonite and zinc compounds derived from it find applications in the chemical industry. Zinc oxide, which can be obtained from smithsonite, is utilized as a white pigment in paints, rubber, ceramics, and cosmetics. It is also used in the production of zinc stearate, a lubricant and release agent, as well as in the formulation of various other chemicals.
- Electrical and Electronic Applications: Smithsonite indirectly contributes to the electrical and electronic industries through its role in zinc production. Zinc is a key component in the manufacture of batteries, including zinc-carbon and alkaline batteries, due to its ability to provide electrical conductivity and long-lasting power.
- Mineral Collecting and Jewelry: Beyond its industrial uses, smithsonite is highly valued in the world of mineral collecting. Specimens with vibrant colors and unique crystal habits are sought after by collectors for their aesthetic appeal. Smithsonite can also be cut and polished to create attractive cabochons, beads, and other gemstone forms for use in jewelry.
- Decorative Stone: Smithsonite, particularly specimens with eye-catching colors and patterns, can be used as a decorative stone in architecture and interior design. It is often incorporated into countertops, wall claddings, and other ornamental applications.
It’s worth noting that while smithsonite has industrial applications, its availability as an ore of zinc may vary depending on the specific geological deposits and market conditions. Additionally, due to its relative softness, proper care is required when using smithsonite in commercial applications to prevent scratching or damage.
Smithsonite is a gemstone
Smithsonite, with its attractive colors and unique appearance, is also valued and used as a gemstone in the world of jewelry. Here’s some information about Smithsonite as a gemstone:
- Color and Varieties: Smithsonite gemstones come in a wide range of colors, including blue, green, pink, gray, and white. The most sought-after colors are vibrant blues and greens, resembling turquoise or seafoam. Multicolored specimens with banding or patterns are also highly prized. The various colors of smithsonite make it a versatile gemstone for jewelry design.
- Durability: Smithsonite has a relatively low hardness of 4 to 5 on the Mohs scale, which means it is softer than many other gemstones commonly used in jewelry. As a result, care must be taken to prevent scratching or damage. It is best suited for jewelry pieces that won’t be subjected to excessive wear or potential impact.
- Cutting and Polishing: Smithsonite gemstones are typically cut and polished into cabochons or beads to showcase their unique colors and patterns. The cabochon cut, with its smooth, rounded surface, is particularly popular for smithsonite. The gemstones can also be carved into various shapes, enhancing their visual appeal.
- Jewelry Applications: Smithsonite gemstones are used in various types of jewelry, including rings, pendants, earrings, bracelets, and necklaces. Due to its relatively soft nature, smithsonite is often set in protective settings, such as bezel or halo settings, to provide extra security and minimize the risk of damage.
- Collectibility and Rarity: Fine-quality smithsonite gemstones, especially those with intense and vibrant colors, are highly collectible and sought after by gemstone enthusiasts. However, large, high-quality smithsonite specimens are relatively rare, which contributes to their value and desirability.
- Care and Maintenance: Due to its relative softness, smithsonite gemstones require gentle care and protection to preserve their beauty. It is recommended to avoid exposing them to harsh chemicals, ultrasonic cleaning, or prolonged exposure to sunlight, as these factors can potentially damage the stone. Instead, cleaning with mild soap and a soft cloth is generally recommended.
Overall, smithsonite’s unique colors and patterns, combined with its relative scarcity, make it a prized gemstone for jewelry connoisseurs and collectors. Its distinctive appearance allows for unique and eye-catching jewelry designs, providing an alternative to more common gemstones.
FAQs
What is smithsonite?
Smithsonite is a mineral composed of zinc carbonate. It is also known as zinc spar or calamine.
How does smithsonite get its name?
Smithsonite is named after James Smithson, an English scientist and benefactor who founded the Smithsonian Institution.
What colors does smithsonite come in?
Smithsonite occurs in a wide range of colors, including blue, green, pink, gray, and white. Multicolored specimens are also found.
Where is smithsonite found?
Smithsonite is found in various countries around the world, including the United States, Mexico, Australia, Namibia, and Greece, among others.
How is smithsonite formed?
Smithsonite is formed as a secondary mineral through the alteration of primary zinc minerals, typically in oxidized zinc ore deposits.
What is the hardness of smithsonite?
Smithsonite has a hardness of 4 to 5 on the Mohs scale, making it relatively soft compared to many other gemstones.
Can smithsonite be used in jewelry?
Yes, smithsonite is used as a gemstone in jewelry. It is often cut into cabochons or beads to showcase its colors and patterns.
Is smithsonite a birthstone?
Smithsonite is not commonly recognized as a birthstone, but its blue varieties can serve as an alternative to turquoise, which is a December birthstone.
How should smithsonite be cared for?
Smithsonite should be handled with care due to its relatively soft nature. It is best to avoid exposure to harsh chemicals and ultrasonic cleaning. Cleaning with mild soap and a soft cloth is recommended.