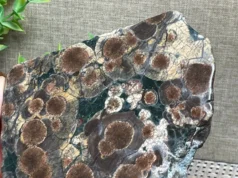
Willemite is a mineral with the chemical formula Zn2SiO4. It is a zinc silicate and is known for its unique fluorescence properties, making it a fascinating material both in the field of mineralogy and as a fluorescent collector’s gem. In this introduction, we’ll explore its definition, crystal structure, and historical background.



Willemite is a rare mineral that consists of zinc, silicon, and oxygen atoms arranged in a specific crystal structure. Its chemical formula is Zn2SiO4, which means that for every two atoms of zinc, there is one atom of silicon and four atoms of oxygen bonded together in a structured lattice.
Crystal Structure: Willemite belongs to the hexagonal crystal system. Its crystal structure is typically classified as hexagonal prismatic, and it forms hexagonal prismatic crystals. The atoms in the crystal lattice are arranged in a way that gives willemite its unique optical properties, including its fluorescence when exposed to ultraviolet (UV) light.
Historical Background and Discovery: Willemite was first discovered in 1830 in the Roughton Gill Mine in Caldbeck Fells, Cumbria, England. It was named after William I of the Netherlands, who was king at the time of its discovery. The mineral gained popularity among mineral collectors for its striking green fluorescence under UV light.
The fluorescence of willemite is a result of trace impurities within the crystal lattice, particularly small amounts of manganese. When these impurities are exposed to UV light, they emit visible light, creating the distinctive green glow that willemite is known for. This property made willemite a sought-after collector’s item, and specimens with intense fluorescence are highly prized.
In addition to its fluorescent properties, willemite is also found in a range of colors, including green, yellow, brown, and rarely, colorless. It can occur in various geological settings, often associated with other zinc minerals such as franklinite and zincite.
Willemite has applications beyond its role as a collector’s gem. It is sometimes used as an ore of zinc, an important industrial metal, and it can also serve as a phosphor in certain types of fluorescent lights and X-ray screens due to its ability to emit light when exposed to ionizing radiation.
In summary, willemite is a unique mineral with a hexagonal crystal structure that is renowned for its striking fluorescence under UV light. Its historical discovery in England in the 19th century and its continued fascination among mineral enthusiasts have made it a notable and intriguing mineral specimen.

Physical Properties
- Color: Willemite can exhibit a range of colors, including green (the most common), yellow, brown, and rarely, colorless. The green color is often due to the presence of trace amounts of manganese as an impurity.
- Luster: It typically has a vitreous (glassy) to sub-adamantine (nearly diamond-like) luster, giving it a shiny appearance when polished.
- Transparency: Willemite can be transparent to translucent, allowing light to pass through to varying degrees, depending on its purity and impurities.
- Crystal Habit: It commonly forms hexagonal prismatic crystals, but it can also occur in granular, massive, or botryoidal forms. Crystals are often elongated, and they may be well-formed or somewhat irregular.
- Cleavage: Willemite exhibits perfect cleavage in one direction parallel to its basal plane. This means that it can be easily split into thin, flat sheets along this plane.
- Hardness: It has a hardness of approximately 5.5 on the Mohs scale, which makes it moderately hard but not as hard as some other gemstones and minerals.
- Density: The density of willemite typically falls in the range of 3.87 to 4.08 grams per cubic centimeter, depending on its composition.
Chemical Properties
- Chemical Formula: The chemical formula of willemite is Zn2SiO4, indicating that it is composed of zinc (Zn), silicon (Si), and oxygen (O) atoms in a specific ratio.
- Composition: Willemite is primarily composed of zinc, silicon, and oxygen. The green color seen in willemite is often the result of manganese (Mn) impurities.
- Trace Elements: Other trace elements and impurities, such as iron (Fe), calcium (Ca), and magnesium (Mg), can also be present in willemite, influencing its color and properties.
Optical Properties
- Fluorescence: One of the most famous optical properties of willemite is its strong fluorescence. When exposed to ultraviolet (UV) light, willemite emits a bright green or yellow-green glow. This property is due to the presence of manganese impurities within the crystal lattice.
- Birefringence: Willemite is birefringent, meaning it can split light into two polarized rays as it passes through the crystal. This property can be observed under a polarizing microscope and is used in mineral identification.
- Refractive Index: The refractive index of willemite varies depending on the composition and color. It typically falls in the range of 1.68 to 1.80 for the green variety, but it can be higher or lower for different colors.
- Dispersion: Willemite exhibits moderate dispersion, which means it can separate light into its spectral colors, producing some fire or color flashes when viewed under certain lighting conditions.
Overall, willemite’s physical, chemical, and optical properties, including its unique fluorescence and crystal structure, contribute to its appeal both as a mineral specimen and a gemstone.
Willemite Occurrence and Formation

Willemite occurs in a variety of geological settings and forms through several different processes. Its occurrence and formation are influenced by factors such as the presence of zinc-rich minerals, the availability of silicon, and the influence of hydrothermal and metamorphic processes. Here’s a closer look at how and where willemite is found:
- One common occurrence of willemite is in hydrothermal mineral deposits. These deposits form when hot, mineral-rich fluids circulate through fractures and cavities in the Earth’s crust. These fluids often contain dissolved zinc, which can precipitate out as willemite when conditions are right.
- Willemite can be found in association with other zinc minerals like sphalerite and galena in these hydrothermal deposits.
2. Contact Metamorphism:
- Willemite can also form during contact metamorphism, a process in which rocks are altered due to the heat and chemical changes associated with the intrusion of molten magma. In this setting, willemite may develop from the alteration of zinc-rich carbonate rocks in the presence of silica.
3. Franklinite-Zincite-Willemite Ore Bodies:
- One of the most famous occurrences of willemite is in the Franklin-Sterling Hill mining district in New Jersey, USA. Here, willemite is found in significant quantities in ore bodies alongside other zinc minerals like franklinite and zincite. These ore bodies are known for their exceptional fluorescence.
4. Skarn Deposits:
- Willemite can be associated with skarn deposits, which form near the contact between igneous intrusions and carbonate rocks. The interaction between the intruding magma and the carbonate rocks can lead to the formation of willemite, among other minerals.
5. Secondary Deposits:
- In some cases, willemite may form as a secondary mineral through the alteration of pre-existing minerals, such as sphalerite or zincite, under the influence of weathering and groundwater.
6. Collectors’ Deposits:
- Collectors of fluorescent minerals often discover willemite in abandoned mines, quarries, or rock outcrops. These specimens are highly sought after for their intense green fluorescence under ultraviolet light.
It’s important to note that willemite’s unique fluorescence is often a result of trace impurities within the crystal lattice, particularly small amounts of manganese. The presence of manganese impurities causes the willemite crystals to emit green or yellow-green light when exposed to ultraviolet (UV) light, adding to its allure among mineral enthusiasts.
In summary, willemite occurs in a range of geological settings, including hydrothermal deposits, contact metamorphic environments, skarn deposits, and secondary deposits. Its formation is influenced by the availability of zinc and silicon, as well as the influence of various geological processes. Its distinctive fluorescence makes it a prized mineral specimen in the world of mineral collecting.
Mining Location, Deposits

Willemite is not typically mined on a large scale for industrial or commercial purposes like many other minerals. Instead, willemite is primarily sought after by mineral collectors and enthusiasts due to its striking green fluorescence under ultraviolet (UV) light. As a result, willemite is most commonly found in specific geological settings and mining locations known for producing exceptional specimens for collectors. The Franklin-Sterling Hill mining district in New Jersey, USA, is one of the most renowned locations for willemite mining and collection. Here’s more information about it:
Location: Franklin-Sterling Hill Mining District, New Jersey, USA:
- The Franklin-Sterling Hill mining district, located in Sussex County, New Jersey, is world-famous for its deposits of fluorescent minerals, including willemite.
- The district is home to a unique geological setting known as the Franklin Marble, which contains a rich variety of zinc minerals, including willemite, franklinite, and zincite.
- Willemite specimens from this area are highly prized by collectors for their intense green fluorescence when exposed to UV light.
- The district’s mines, including the Franklin Mine and the Sterling Hill Mine, have been historically important sources of fluorescent minerals, and they have yielded some of the most beautiful willemite specimens in the world.
It’s important to note that while the Franklin-Sterling Hill mining district is a significant location for collecting willemite specimens, these minerals are not typically mined for industrial purposes. The primary focus of activity in this area is mineral collecting and research rather than large-scale mining operations.
Additionally, willemite may be found in smaller quantities in other geological settings where zinc and silicate minerals are present, but the intense fluorescence that makes it famous is a distinctive feature of specimens from the Franklin-Sterling Hill district. Collectors and mineral enthusiasts often visit this area to search for willemite and other fluorescent minerals.
Uses and Applications

Willemite, while not a widely used industrial mineral, has some niche applications and uses primarily driven by its unique optical properties. Here are some of the main uses and applications of willemite:
- Mineral Collecting: One of the primary uses of willemite is in the field of mineral collecting. It is highly prized by mineral enthusiasts and collectors for its intense green fluorescence when exposed to ultraviolet (UV) light. Specimens of willemite are sought after for their aesthetic appeal and rarity.
- Gem and Jewelry Industry: Although willemite is not a mainstream gemstone, it can be cut and polished into cabochons or faceted gems. Jewelry designers and collectors may use it for its attractive green color and fluorescence. However, it’s not as popular as other gemstones due to its relative softness and scarcity in gem-quality specimens.
- Phosphors: Willemite has been used as a phosphor in the past, primarily in older fluorescent light tubes and cathode ray tubes (CRTs). Its ability to emit visible light when exposed to ionizing radiation, such as X-rays, made it useful for creating fluorescent screens in these devices. However, modern technology has largely replaced willemite with more efficient phosphors.
- Scientific Research: Willemite’s unique fluorescence properties have made it valuable in scientific research, particularly in the study of fluorescence and luminescence. Researchers may use willemite specimens as reference materials or in experiments related to fluorescence spectroscopy.
- Mineralogy and Geology Education: Willemite is a notable mineral in the field of mineralogy and geology education. It is used as a teaching tool to demonstrate concepts related to mineral identification, fluorescence, and crystallography. Its distinctive green fluorescence makes it engaging for educational purposes.
- Art and Crafts: Some artisans and craft enthusiasts may incorporate willemite specimens into their art and craft projects, especially when looking for materials that exhibit unique optical properties.
It’s essential to note that willemite’s limited commercial applications are primarily due to its rarity and the availability of more suitable materials for specific purposes. While it may not have widespread industrial uses, its beauty and uniqueness have made it a coveted mineral in the world of mineral collecting and a subject of interest in various scientific and educational fields.
Varieties and Related Minerals
Willemite, a zinc silicate mineral, can exhibit a range of varieties and is often associated with other minerals in geological settings. These related minerals can have varying compositions and characteristics. Here are some notable willemite varieties and related minerals:
Varieties of Willemite:
- Franklinite-Willemite Series: In the Franklin-Sterling Hill mining district of New Jersey, USA, willemite is commonly associated with franklinite and zincite. This trio of minerals is often found together and is collectively referred to as the Franklinite-Willemite Series. Franklinite is a zinc iron oxide mineral, and zincite is a zinc oxide mineral. These minerals often occur in association with willemite and are known for their exceptional fluorescence.
- Calamine: Calamine is a variety of willemite that is typically massive and usually contains some iron impurities. It can vary in color from white to pink or brown. Historically, calamine has been used in the production of zinc oxide, and it has been used in medicinal lotions and ointments due to its soothing properties.
- Troostite: Troostite is a variety of willemite that contains significant amounts of iron. It is typically yellowish to reddish-brown in color and may exhibit a brown streak. Troostite is named after the Dutch chemist J.B. Troost.
Related Minerals:
- Zincite: Zincite is a zinc oxide mineral often found in association with willemite, especially in the Franklin-Sterling Hill mining district. It can exhibit a range of colors, including red, orange, and brown. Zincite is known for its rarity and vibrant colors.
- Franklinite: As mentioned earlier, franklinite is commonly found alongside willemite in the Franklin-Sterling Hill mining district. It is a zinc iron oxide mineral and can be black to brownish-black in color. Franklinite is a key member of the Franklinite-Willemite Series and is known for its magnetic properties.
- Hemimorphite: Hemimorphite is a zinc silicate mineral that can sometimes be associated with willemite deposits. It is typically colorless, white, or blue, and it often forms botryoidal or stalactitic masses.
- Smithsonite: Smithsonite is a secondary zinc carbonate mineral that can occasionally occur alongside willemite. It can exhibit a variety of colors, including white, green, blue, pink, and brown. Smithsonite has been used as an ore of zinc in some mining operations.
- Sphalerite: Sphalerite is a zinc sulfide mineral and one of the most common ore minerals for zinc. While it is not a direct variety of willemite, it is often found in the same geological environments. Sphalerite can vary in color from yellow to brown to black.
These varieties and related minerals highlight the diverse geological settings and compositions in which willemite can be found. The presence of impurities and the association with other minerals contribute to the range of colors and properties observed in willemite specimens from different locations.