Serpentine is the common name of a group of minerals. Apart from the main members of Antigorite and Chrysotile, there is usually no distinction between individual members except for scientific study and classification. Antigorite generally represents more solid forms, and Chrysotile often represents fibrous forms, especially asbestos. Chrysotile divides the four-membered mineral into its subclass with its crystallization, and the clinocotylot is the most common form of Chrysotile to date.
In this formula, X will be one of the following metals: magnesium, iron, nickel, aluminum, zinc, or manganese; and, Y will be silicon, aluminum, or iron. The appropriate generalized formula is thus
(Mg,Fe,Ni, Mn,Zn)2-3(Si,Al,Fe)2O5(OH)4.
Serpentine Formation
Serpentine minerals, peridotite, dunite and different ultramafic rocks are exposed to hydrothermal metamorphism. Ultramafic rocks are rare on the Earth level, but abundant in the ocean mohounda, at the boundary between the bottom of the ocean crust and the upper mantle.
They are metamorphosed in convergent restrictions where the ocean plate is inserted into the mantle. This is their exposure to hydrothermal metamorphism. The water source for this method is the sea water in the rocks and sediments of the ocean plate.
Serpentine Physical Properties
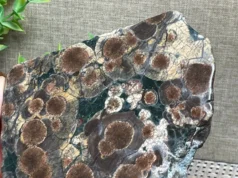
The most obvious physical properties of serpentine are its green color, patterned appearance, and slippery feel. These remind the observer of a snake and that is where the name “serpentine” was derived.
| Chemical Classification | Silicate |
| Color | Usually various shades of green, but can be yellow, black, white, and other colors. |
| Streak | White |
| Luster | Greasy or waxy |
| Diaphaneity | Translucent to opaque, rarely transparent |
| Cleavage | Poor to perfect |
| Mohs Hardness | Variable between 3 and 6 |
| Specific Gravity | 2.5 to 2.6 |
| Diagnostic Properties | Color, luster, fibrous habit, hardness, slippery feel |
| Chemical Composition | (Mg,Fe,Ni,Al,Zn,Mn)2-3(Si,Al,Fe)2O5(OH)4 |
| Crystal System | Most serpentine minerals are monoclinic. |
| Uses | A source of asbestos, architectural stone, ornamental stone, gem material. |

Serpentine Optical Properties

| Formula | Mg3Si2O5(OH)4 Very minor substitution of Al for Si, and of Fe and Al for Mg. |
| Crystal System | Monoclinic |
| Crystal Habit | Crysotile: Fibrous, elongated, and parallel to crystallographic axis a Lizardite and antigorite: flat, tabular crystals |
| Cleavage | Chrysotile: fibrous Lizardite: basal cleavage Antigortie: perfect {001} |
| Color/Pleochroism | Green in thin section |
| Optic Sign | Biaxial (-) |
| 2V | highly variable, may be sensibly uniaxial |
| Optic Orientation | Slow ray vibration direction is typically parallel to the length of fibers in chrysotile giving it parallel extinction. For antigorite – Optic plane is perp to (010). X=c, Y=b, Z=a |
| Refractive Indices alpha = beta = gamma = | 1.538-1.567 ~1.566 1.545-1.574 |
| Max Birefringence | .001-.010 |
| Elongation | Chrysotile is length-slow |
| Extinction | Parallel to fibres, cleavage or crystal edge. |
| Dispersion | r > v for antigorite |
| Distinguishing Features | With the exception of cross-fibers of chrystolite in veins, the varietites of serpentine cannot be distinguished without X-ray diffraction or other techniques. |
| Associated Minerals | talc, calcite, brucite, chlorite, and chromite. |
| Editors | Emilie Flemer (’01), Jennifer Unis (’01), Rebecca-Ellen Farrell (’03), Liz Hogan (’04), Sofia Johnson (’19) |
Serpentine Uses
- Serpentine has been used as an architectural stone for lots of years. It is available in a huge type of inexperienced and greenish shades, often has an attractive sample, works without difficulty, and polishes to a pleasant luster. It has a Mohs hardness of three to six that’s softer than granite, and usually harder than most marble. This low hardness limits its appropriate use to surfaces so that it will not get hold of abrasion or put on, such as facing stone, wall tiles, mantles, and window sills.
- Some varieties of serpentine have a fibrous habit. These fibers resist the transfer of heat, do not burn, and serve as excellent insulators. The serpentine mineral chrysotile is common, found in many parts of the world, is easily mined, and can be processed to recover the heat-resistant fibers.
- Attractive serpentine can be cut into a wide variety of gemstones. It is most often cut into cabochons
- Some varieties of serpentine can be carved into beautiful stone sculptures. Fine-grained, translucent material with a uniform texture and without voids and fractures is preferred. Serpentine is relatively soft and carves easily. It also accepts a nice polish.
- They usually display a range of green, yellow, and black colors and often have magnetite, chromite, or other minerals as interesting inclusions. The lower left side of the green and black cabochon in the center of the photo on this page contains enough included magnetite that the cab can be moved with a small hand magnet.
- Serpentinite rock units have been considered as repositories for the disposal of waste carbon dioxide produced when fossil fuels are burned. Injecting carbon dioxide into subsurface rock units in the presence of water can produce magnesium carbonate and quartz in an exothermic reaction similar to the one shown below.