
Orpiment is a rare mineral composed of arsenic trisulfide, with the chemical formula As2S3. It is known for its distinctive bright yellow to orange-yellow color and has been used as a pigment in painting and for various purposes throughout history. Here are some key points about orpiment:
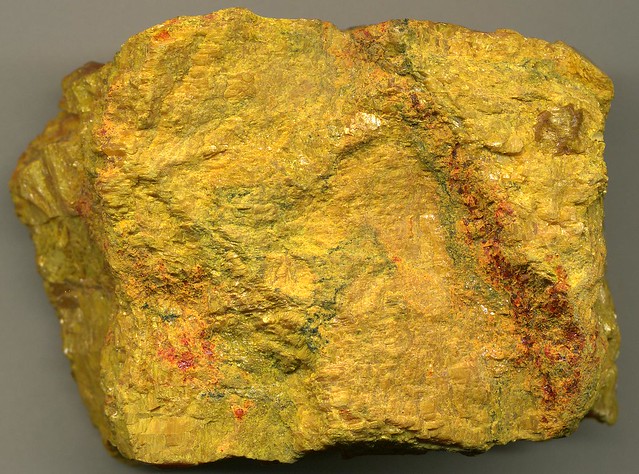


Appearance: Orpiment typically forms in monoclinic crystals, but it can also appear in massive or granular forms. Its vibrant yellow or orange-yellow color makes it easily recognizable.
Occurrence: Orpiment is found in hydrothermal deposits associated with other sulfide minerals and can be associated with realgar, another arsenic sulfide mineral.
Historical Uses: Orpiment has a long history of use in art and ancient civilizations. It was used as a pigment in painting, particularly in ancient Egypt and China. Its use, however, declined due to its toxic nature.
Toxicity: Orpiment contains arsenic, a highly toxic element. Inhaling or ingesting arsenic compounds can lead to severe health problems, including death. Due to its toxicity, orpiment is no longer used in art or industry.
Mineral Collecting: Despite its toxicity, orpiment is collected by mineral enthusiasts and collectors for its striking color and crystal formations. However, collectors must handle it with care and take necessary safety precautions.
Geological Significance: Orpiment can be an indicator of gold mineralization in some geological settings. In certain regions, the presence of orpiment might be associated with gold deposits.
Chemical Properties: Orpiment is composed of two arsenic atoms bonded to three sulfur atoms (As2S3). It has a relatively low hardness on the Mohs scale, making it relatively easy to scratch.
Color Variations: The color of orpiment can vary depending on impurities. Pure orpiment is a bright yellow, while impurities can give it an orange or red tint.
Crystalline Structure: Orpiment has a monoclinic crystal structure, which means its crystals have non-right angles in their internal arrangement.
Synthetic Orpiment: In modern times, synthetic orpiment can be produced for research and industrial purposes without the health hazards associated with natural orpiment.
Name: From the Latin auripigmentum, golden paint, in allusion to the color.
Cell Data: Space Group: P21/n. a = 11.475(5) b = 9.577(4) c = 4.256(2) β = 90◦41(5)0 Z=4
X-ray Powder Pattern: Baia Sprie (Fels˝ob´anya), Romania. 4.85 (100), 4.02 (50), 2.47 (40), 1.755 (40), 3.22 (30), 2.79 (30), 2.72 (30)
Association: Stibnite, realgar, arsenic, calcite, barite, gypsum.
Crystallography: Monoclinic; prismatic. Crystals small, tabular or short prismatic, and rarely distinct. Usually in foliated or columnar masses
Due to its toxicity, orpiment is not commonly encountered in everyday life, but it remains an interesting and sought-after mineral for collectors and researchers due to its unique properties and history.

Chemical Properties of Orpiment
| Chemical Classification | Sulfide mineral |
| Chemical Composition | As2S3 |
Physical Properties of Orpiment
| Color | Lemon-yellow to golden or brownish yellow |
| Streak | Pale lemon-yellow |
| Luster | Resinous, pearly on cleavage surface |
| Cleavage | Perfect Perfect {010} imperfect {100} |
| Diaphaneity | Transparent |
| Mohs Hardness | 1.5 – 2 |
| Crystal System | Monoclinic |
| Tenacity | Sectile |
| Density | 3.49 |
Optical Properties of Orpiment
| Type | Anisotropic |
| Color / Pleochroism | Strong |
| 2V: | Measured: 30° to 76°, Calculated: 62° |
| RI values: | nα = 2.400 nβ = 2.810 nγ = 3.020 |
| Twinning | On {100} |
| Optic Sign | Biaxial (-) |
| Birefringence | δ = 0.620 |
| Relief | Very High |
| Dispersion: | relatively strong r > v |
Orpiment Occurrence and Formation

Orpiment primarily occurs in hydrothermal mineral deposits, and its formation is closely tied to specific geological conditions. Here’s a more detailed explanation of the occurrence and formation of orpiment:
Occurrence:
- Hydrothermal Deposits: Orpiment is most commonly found in hydrothermal mineral deposits. These deposits are formed when hot, mineral-rich fluids circulate through fractures and cavities in rocks. The fluids are often associated with volcanic or magmatic activity deep within the Earth’s crust.
- Sulfide Minerals Association: Orpiment is often associated with other sulfide minerals, including realgar (another arsenic sulfide mineral), pyrite, and various sulfides of other metals. These minerals can form together in the same hydrothermal veins or deposits.
- Specific Geological Settings: Orpiment tends to occur in specific geological settings, such as volcanic rocks, hot springs, and geothermal areas. It can also be found in sedimentary rocks in some cases.
Formation:
The formation of orpiment involves a series of geological processes:
- Source of Arsenic and Sulfur: The essential elements for orpiment, arsenic, and sulfur, must be present in the geological environment. Arsenic is often introduced into the system through magmatic or hydrothermal processes, while sulfur may come from various sources, including hydrothermal fluids and rocks.
- Hydrothermal Fluids: Hot, mineral-rich fluids (hydrothermal fluids) rise through fractures and fissures in the Earth’s crust. These fluids can have temperatures ranging from moderately warm to very hot.
- Precipitation: As the hydrothermal fluids move through the rock layers, they encounter conditions that lead to the precipitation of minerals. Orpiment forms when the concentration of arsenic and sulfur in the fluid reaches a point where they can react and precipitate as orpiment crystals.
- Temperature and Pressure Changes: Changes in temperature, pressure, and chemical conditions within the hydrothermal system play a crucial role in the formation of orpiment. These changes can trigger the precipitation of minerals, including orpiment, from the fluid.
- Crystallization: Orpiment crystals can grow over time as more arsenic and sulfur are supplied by the hydrothermal fluids. The resulting crystals can vary in size and quality depending on the specific conditions of the deposit.
- Associated Minerals: Orpiment is often found alongside other minerals, such as realgar, due to the similar geological processes that lead to their formation.
It’s important to note that while orpiment is visually striking due to its vibrant yellow color, it is highly toxic due to its arsenic content. Therefore, anyone involved in the exploration, collection, or study of orpiment-containing deposits should exercise caution and follow appropriate safety guidelines to minimize exposure to the toxic mineral.
Application and Uses Areas

Orpiment, which is composed of arsenic trisulfide (As2S3), has limited applications and uses due to its toxicity. Historically, it was primarily used as a pigment in art and decoration, but its use has significantly declined in modern times because of its health hazards. Here are the historical and limited modern applications and use areas of orpiment:
- Historical Use as a Pigment: Orpiment was highly valued as a yellow pigment in ancient art, particularly in ancient Egypt and China. It was used for painting murals, manuscripts, and decorative objects. However, its use declined as the toxic nature of arsenic became better understood.
- Ink and Dye: Orpiment was occasionally used in the production of yellow inks and dyes in historical contexts. Again, this use has diminished due to health concerns.
- Pyrotechnics: Orpiment was historically used in fireworks and pyrotechnics to create yellow and white flames. However, safer alternatives are now preferred for such applications.
- Alchemical and Medicinal Uses: In ancient times, orpiment was used in alchemical practices, but these were often based on mystical beliefs and superstitions. It was also used in traditional Chinese medicine, but its toxic properties have led to its replacement with safer alternatives.
- Mineral Collecting: Orpiment is occasionally collected by mineral enthusiasts and collectors for its striking yellow color and crystal formations. Collectors must handle it with great care and follow safety precautions due to its toxicity.
- Industrial Applications: Orpiment has limited modern industrial applications. It can be used in the manufacturing of certain types of glass, particularly yellow or yellowish-green glass. However, alternatives that do not contain toxic arsenic are preferred.
- Geological Significance: In a geological context, the presence of orpiment in specific rock formations can sometimes be an indicator of certain geological conditions, such as hydrothermal mineralization. In some cases, orpiment’s presence may suggest the potential for valuable mineral deposits like gold.
It’s important to emphasize that the use of orpiment has diminished significantly in modern times due to its toxicity. The health risks associated with exposure to arsenic, a component of orpiment, have led to the discontinuation of its use in many applications. Safer and less toxic alternatives have been developed for various purposes, particularly in the fields of art, chemistry, and industry.
Mining Sources, Distribution

Orpiment is primarily mined from geological formations where it occurs naturally. Its distribution is closely linked to specific geological conditions and the presence of arsenic and sulfur-rich minerals. Here’s a closer look at the mining sources and distribution of orpiment:
Mining Sources:
- Hydrothermal Veins: Orpiment is commonly found in hydrothermal mineral deposits. These deposits are formed when hot, mineral-rich fluids circulate through fractures and fissures in rocks. Orpiment can precipitate from these hydrothermal fluids under the right conditions.
- Volcanic Environments: Orpiment can be associated with volcanic rocks and geothermal areas. Volcanic processes can introduce arsenic and sulfur into the geological environment, which are necessary components for orpiment formation.
- Sedimentary Deposits: In some cases, orpiment can also occur in sedimentary rock formations. These deposits are typically formed through the alteration of pre-existing minerals and the deposition of orpiment from fluids that have leached arsenic and sulfur from other sources.
- Associated Minerals: Orpiment is often found alongside other minerals, such as realgar (another arsenic sulfide mineral) and various sulfides of other metals. These associated minerals can occur in the same geological settings and are often mined together.
Distribution:
Orpiment is found in various parts of the world, but its distribution is not widespread due to its specific geological requirements. Some regions known for significant orpiment deposits include:
- China: China has historically been a major source of orpiment. It has been mined in various provinces, including Hunan, Hubei, and Yunnan. The Chinese name for orpiment, “Yellow Arsenic,” reflects its historical significance in the country.
- Peru: Orpiment has also been mined in Peru, where it is associated with volcanic and hydrothermal deposits in the Andes Mountains.
- Romania: Orpiment deposits have been reported in certain regions of Romania, often in association with other sulfide minerals.
- Turkey: Turkey is another country with orpiment deposits, and it has been mined in the past, although production levels may vary over time.
- Other Locations: Orpiment can be found in other countries and regions with suitable geological conditions, but its occurrence is generally less common compared to other minerals.
It’s important to note that the mining and use of orpiment have significantly declined in modern times due to its toxic nature. Strict safety measures and precautions are necessary when working with orpiment-containing deposits to minimize exposure to the harmful effects of arsenic. Moreover, the availability of safer alternatives for various industrial and artistic purposes has led to a reduced demand for orpiment.
References
- Bonewitz, R. (2012). Rocks and minerals. 2nd ed. London: DK Publishing.
- Handbookofmineralogy.org. (2019). Handbook of Mineralogy. [online] Available at: http://www.handbookofmineralogy.org [Accessed 4 Mar. 2019].
- Mindat.org. (2019). Orpiment: Mineral information, data and localities.. [online] Available at: https://www.mindat.org/ [Accessed. 2019].
- Smith.edu. (2019). Geosciences | Smith College. [online] Available at: https://www.smith.edu/academics/geosciences [Accessed 15 Mar. 2019].




























