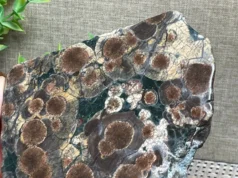
Zincite is a mineral composed primarily of zinc oxide (ZnO), often with trace amounts of manganese and other elements. It typically occurs in bright red, orange, or yellowish-orange colors, although it can also be found in green and brown hues. The name “zincite” is derived from its composition, with “zinc” referring to its primary element and “-ite” denoting its status as a mineral.

Zincite is primarily known for its vibrant coloration and is often prized by mineral collectors and lapidaries for its aesthetic appeal. It forms in various geological environments, including zinc ore deposits and metamorphic rocks, and can sometimes be found as crystals or in granular form.
In addition to its ornamental uses, zincite also has industrial applications. Zinc oxide, the main component of zincite, is widely used in the production of rubber, ceramics, paints, cosmetics, and various other products. It is valued for its ability to impart certain properties, such as UV protection and antimicrobial effects, to these materials.
Overall, zincite is a fascinating mineral with both aesthetic and practical significance, embodying the unique properties and applications of zinc oxide in natural form.
History of discovery and early uses

Zincite’s history dates back to the early 19th century when it was first discovered and recognized as a distinct mineral. Here’s a brief overview of its discovery and early uses:
- Discovery: Zincite was first identified as a mineral in 1810 by the German mineralogist and geologist Franz Ludwig von Kobell. He found it in association with zinc ore deposits in Poland. Kobell named the mineral “zincite” due to its composition primarily consisting of zinc oxide.
- Early Uses: In its early history, zincite was primarily valued for its vibrant colors and aesthetic appeal. Mineral collectors prized specimens for their beauty, and lapidaries sometimes used the material to create ornamental objects and jewelry.
- Industrial Applications: As the industrial revolution progressed, zincite found practical applications beyond its ornamental uses. Zinc oxide, the main component of zincite, was recognized for its various properties, including its ability to act as a pigment in paints and ceramics. Its ability to absorb ultraviolet (UV) light made it valuable in the production of sunscreens and other UV-blocking products. Additionally, zinc oxide’s antimicrobial properties led to its use in ointments and creams for treating skin conditions.
- Scientific Interest: Beyond its commercial applications, zincite has also attracted scientific interest due to its unique crystal structure and properties. Researchers have studied zincite’s structure and behavior under different conditions to gain insights into its optical, electrical, and magnetic properties.
Overall, zincite’s discovery marked the beginning of its journey from a mineral of aesthetic value to one with diverse industrial and scientific applications. Its history reflects the intersection of mineralogy, industry, and scientific inquiry over the past two centuries.
Chemical Composition and Structure

Zincite’s chemical composition is primarily zinc oxide (ZnO), with the formula ZnO. However, it may contain trace amounts of other elements, such as manganese (Mn), which can impart different colors to the mineral. The presence of manganese is responsible for zincite’s characteristic red, orange, or yellowish-orange colors.
In terms of structure, zincite crystallizes in the hexagonal system, forming hexagonal prisms or pyramidal crystals. Its crystal structure consists of zinc and oxygen atoms arranged in a repeating pattern. Each zinc atom is surrounded by six oxygen atoms, forming a tetrahedral coordination geometry.
The arrangement of atoms in zincite gives rise to its unique physical and optical properties. For example, zinc oxide is known for its semiconducting properties, making zincite potentially useful in electronic devices. Its optical properties, including its ability to absorb and emit light, have also been studied for applications in optoelectronics and photonics.
Zincite’s crystal structure and composition make it an intriguing subject of study for mineralogists, materials scientists, and engineers interested in understanding and exploiting its properties for various technological applications.
Physical properties

Zincite possesses several distinctive physical properties that contribute to its identification and characterization. Here are some key physical properties of zincite:
- Color: Zincite is known for its vibrant colors, which can range from bright red, orange, and yellowish-orange to green and brown. The specific coloration is often influenced by trace impurities, such as manganese, present within the mineral.
- Luster: The luster of zincite is typically described as adamantine to vitreous, meaning it can exhibit a brilliant, glassy sheen when polished or a reflective, metallic appearance in certain cases.
- Transparency: Zincite can vary in transparency from transparent to translucent. In some cases, particularly when the crystal faces are well-developed, zincite crystals can exhibit excellent transparency, allowing light to pass through with minimal obstruction.
- Crystal Habit: Zincite commonly occurs as hexagonal prismatic crystals or pyramidal forms. These crystals can be well-formed and exhibit distinct crystal faces. However, zincite can also occur in massive, granular, or botryoidal (grape-like) forms.
- Hardness: Zincite has a hardness of around 4 on the Mohs scale, which means it is relatively soft compared to many common minerals. This hardness is comparable to that of materials like fluorite or apatite.
- Cleavage: Zincite typically exhibits no cleavage, meaning it does not break along specific planes of weakness. Instead, it tends to fracture irregularly, producing uneven or conchoidal (shell-like) surfaces.
- Density: The density of zincite varies depending on its composition and degree of crystallinity but generally ranges from 5.6 to 6.0 grams per cubic centimeter (g/cm³). This places zincite at a moderate density compared to other minerals.
These physical properties, along with its chemical composition and crystal structure, contribute to zincite’s distinct appearance and behavior, making it recognizable and valuable to mineralogists, collectors, and researchers alike.
Geological formation and occurrence

Zincite typically forms through various geological processes involving the deposition and alteration of zinc-rich materials. Here’s an overview of its geological formation and occurrence:
- Primary Formation: Zincite can form as a primary mineral in association with zinc ore deposits, particularly those containing sphalerite (zinc sulfide) and other zinc-bearing minerals. These deposits often occur in hydrothermal veins, where hot aqueous solutions rich in zinc migrate through fractures and fissures in the Earth’s crust. Under suitable conditions of temperature and pressure, zinc oxide can precipitate from these solutions, leading to the formation of zincite crystals.
- Secondary Formation: In some cases, zincite may also form as a secondary mineral through weathering and alteration processes. Zinc-containing minerals, such as sphalerite, may undergo chemical weathering, releasing zinc ions into groundwater or surface water. These zinc ions can then react with oxygen to form zinc oxide, which can precipitate as zincite under certain environmental conditions. Secondary zincite deposits may be found in weathered rock formations, soils, or sedimentary environments.
- Metamorphic Formation: Zincite can also occur as a product of metamorphic processes involving the alteration of pre-existing zinc-bearing minerals. During metamorphism, rocks containing zinc-rich minerals may undergo recrystallization and metamorphic reactions, leading to the formation of zincite along with other minerals. Metamorphic zincite deposits may be found in association with metamorphosed sedimentary rocks or in regions undergoing regional metamorphism.
- Associated Minerals: Zincite is often found in association with other zinc-bearing minerals, such as sphalerite, smithsonite (zinc carbonate), and willemite (zinc silicate). Additionally, it may occur alongside minerals containing manganese, which can impart different colors to zincite crystals.
- Geographical Distribution: Zincite deposits can be found worldwide, although significant occurrences are often associated with regions known for zinc mining and mineralization. Some notable locations for zincite include Poland, where it was first discovered, as well as regions in the United States, Australia, Germany, and China.
Overall, zincite’s geological formation involves a combination of primary hydrothermal processes, secondary weathering and alteration, and metamorphic transformations, leading to its occurrence in diverse geological settings around the world.
Major sources and mining locations

Zincite is not typically mined as a primary mineral due to its relatively rare occurrence and its association with other zinc-bearing minerals. Instead, it is often obtained as a byproduct of zinc ore mining and processing. Some of the major sources and mining locations where zincite may be found include:
- Poland: Zincite was first discovered in Poland, particularly in the region of Upper Silesia, which has historically been a significant zinc-producing area. The zincite occurrences in Poland are often associated with zinc ore deposits containing sphalerite and other zinc-bearing minerals.
- United States: Several states in the U.S. have notable zincite occurrences, primarily in regions with active or historic zinc mining operations. States such as New Jersey, New York, Pennsylvania, and Tennessee have produced zincite as a byproduct of zinc mining activities.
- Australia: Australia is a major producer of zinc, with significant zinc mining operations in various states, including Queensland, New South Wales, and Western Australia. Zincite occurrences may be found in association with zinc ore deposits in these regions.
- Germany: Germany has a long history of zinc mining, particularly in regions such as the Harz Mountains. Zincite occurrences have been reported in association with zinc ore deposits in Germany, although they may not be as abundant as in some other regions.
- China: China is one of the largest producers of zinc globally, with zinc mining operations located in various provinces. Zincite occurrences may be found in association with zinc ore deposits in regions such as Yunnan, Sichuan, and Inner Mongolia.
These are just a few examples of major sources and mining locations where zincite may be obtained. It’s important to note that zincite is often a secondary or accessory mineral in zinc ore deposits rather than the primary target of mining operations. As a result, specific occurrences of zincite may vary depending on the geology and mineralization characteristics of each region.
Uses and Applications

Zincite, primarily composed of zinc oxide, has several important uses and applications across various industries. Here are some of the key uses and applications of zincite:
- Pigments: Zinc oxide, the main component of zincite, is widely used as a white pigment in paints, coatings, plastics, and rubber products. It provides excellent opacity, brightness, and UV-blocking properties, making it valuable for both indoor and outdoor applications.
- Cosmetics and Skincare: Zinc oxide is a common ingredient in cosmetics, skincare products, and sunscreens due to its ability to provide broad-spectrum UV protection against both UVA and UVB rays. It is used in formulations such as sunscreens, lotions, creams, and ointments to help prevent sunburn and protect the skin from UV damage.
- Medicinal Applications: Zinc oxide has antimicrobial properties and is used in various medicinal products, including ointments, creams, and bandages, to help treat skin conditions such as diaper rash, minor burns, cuts, and irritations. It acts as a barrier against moisture and irritants while promoting healing.
- Rubber Manufacturing: Zinc oxide is a crucial ingredient in the production of rubber products, including tires, belts, hoses, and footwear. It serves as a reinforcing agent, helping to improve the strength, durability, and elasticity of rubber compounds.
- Ceramics and Glass: Zinc oxide is used as a flux in ceramic and glass manufacturing processes to lower the melting point of materials, improve the homogeneity of glazes, and enhance the transparency and brightness of glass products.
- Electronics and Semiconductors: Zincite, as a natural source of zinc oxide, may have potential applications in electronic devices and semiconductor materials. Zinc oxide is a semiconductor material with properties suitable for use in various electronic components, such as transistors, sensors, and light-emitting diodes (LEDs).
- Pharmaceuticals: Zinc oxide nanoparticles derived from zincite are being researched for potential applications in drug delivery systems, diagnostic imaging, and biomedical devices due to their biocompatibility, stability, and controlled release properties.
These are just a few examples of the diverse uses and applications of zincite and zinc oxide in industry, technology, and everyday products. Its versatile properties make it an essential ingredient in various sectors, from manufacturing and healthcare to consumer goods and electronics.