
Zeolites are a group of naturally occurring minerals and synthetic materials known for their unique crystalline structure and remarkable properties. These minerals are often used in a wide range of industrial applications due to their exceptional adsorption, cation exchange, and molecular sieve capabilities. Zeolites have garnered significant attention for their role in various industries, including petrochemical, environmental, and pharmaceutical sectors.


Zeolites are aluminosilicate minerals with a three-dimensional framework structure. They are characterized by the following properties:
Porosity: Zeolites possess a porous structure with regularly sized channels and cavities. These pores are capable of accommodating various cations, water molecules, and other substances.
Cation Exchange: Zeolites have a high affinity for cations (positively charged ions) and can exchange these cations with others in their surroundings. This property makes them valuable for ion-exchange applications.
Adsorption: Zeolites have a strong adsorption capacity for gases, liquids, and even certain organic molecules. They can selectively trap and release specific substances, making them useful in separation processes.
Molecular Sieve Effect: Zeolites act as molecular sieves, allowing only molecules of a certain size and shape to pass through their pores. This property is essential for separation and purification processes.
Historical Background and Discovery:
Zeolites have a long history, dating back to ancient times when they were used as natural minerals for various purposes, such as water purification and as a drying agent. However, the systematic study of zeolites began in the mid-18th century.
In the 18th century, Swedish mineralogist Axel Fredrik Cronstedt identified a group of minerals known as “zeolites” due to their characteristic boiling stones. He recognized their ability to release water when heated, hence the name “zeolite,” derived from the Greek words “zeo” (to boil) and “lithos” (stone).
Further research on zeolites in the 20th century revealed their unique structural properties and the potential for various industrial applications. This led to the synthesis of synthetic zeolites with tailored properties and improved performance.
Importance and Applications in Various Industries:
Zeolites play a crucial role in several industries due to their versatile properties:
- Petrochemical Industry: Zeolites are used as catalysts in oil refining and petrochemical processes to facilitate the conversion of hydrocarbons. They enable selective cracking, isomerization, and other reactions that enhance the quality and yield of fuels and chemicals.
- Environmental Remediation: Zeolites are employed for water purification and wastewater treatment to remove heavy metals, ammonia, and other contaminants. They can also adsorb and store radioactive waste and help mitigate environmental pollution.
- Detergent Industry: Zeolites are added to detergents as builders to improve cleaning efficiency. They soften water by removing hardness ions (e.g., calcium and magnesium), which can interfere with detergent performance.
- Gas Separation: Zeolites are used for gas separation processes, such as nitrogen and oxygen production, due to their molecular sieve effect. They selectively allow specific gases to pass through their pores.
- Catalysis and Chemical Industry: Zeolites find applications in the chemical industry as catalysts for various reactions, including the synthesis of chemicals and the removal of pollutants.
- Pharmaceuticals and Agriculture: Zeolites have applications in drug delivery systems and as carriers for controlled release of pharmaceuticals and nutrients in agriculture.
In summary, zeolite minerals are remarkable materials with a wide range of applications in diverse industries, thanks to their unique structural and chemical properties. Their ability to adsorb, exchange cations, and act as molecular sieves has made them invaluable in addressing numerous challenges in modern industrial and environmental processes.
Name: The term zeolite was first written in 1756 by the Swedish mineralogist Axel Fredrik Cronstedt. He named the zeolite material from the Greek z (zéō) meaning “stone” to “zeolite”.
Zeolite Crystal Structure
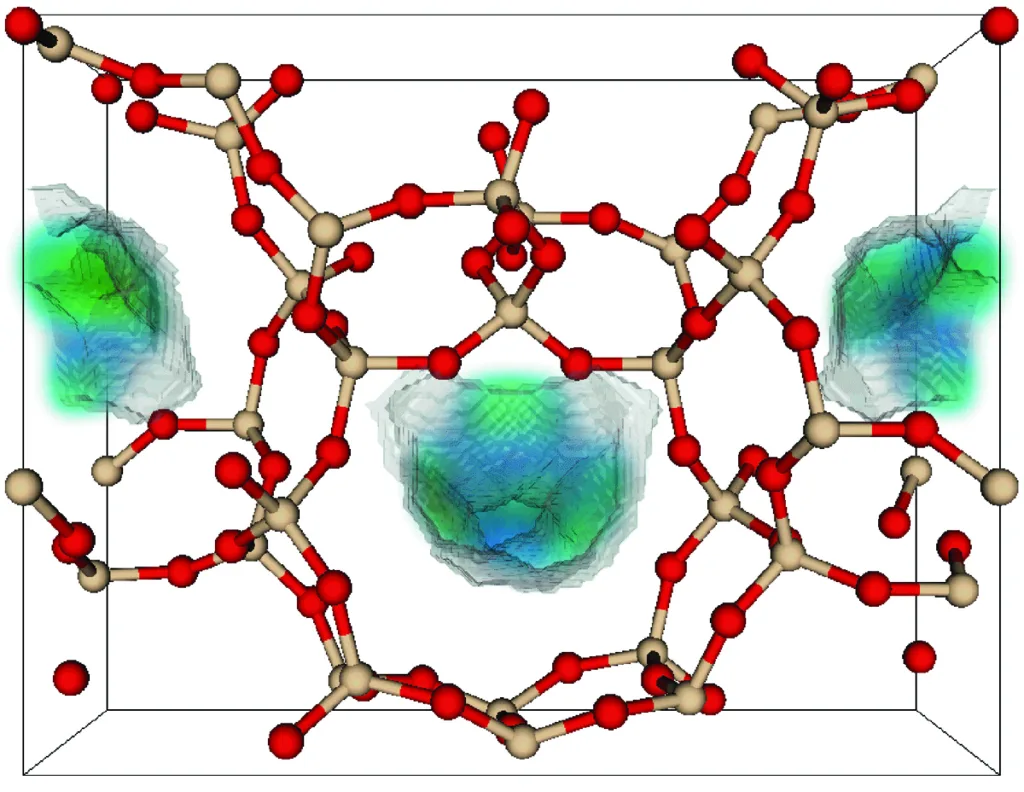
Zeolites are characterized by a highly ordered and crystalline three-dimensional framework structure composed of silicon (Si), aluminum (Al), and oxygen (O) atoms, connected by bridging oxygen atoms. The basic building blocks of zeolite crystal structures are tetrahedral units, which consist of a central silicon or aluminum atom bonded to four oxygen atoms. This tetrahedral arrangement gives zeolites their unique properties, including their porous and cation-exchange capabilities.
Here are the key features of zeolite crystal structures:
- Tetrahedral Units: Each tetrahedral unit in a zeolite structure consists of a central Si or Al atom bonded to four oxygen atoms. These tetrahedra are interconnected to form the framework.
- Framework Structure: Zeolites have an interconnected framework of these tetrahedral units, forming a three-dimensional lattice. This lattice structure results in a network of channels and cavities within the crystal.
- Pores and Channels: The framework structure of zeolites creates a system of pores and channels of uniform size and shape throughout the crystal. These channels provide the zeolite with its exceptional porosity and adsorption properties.
- Cation Sites: Within the channels and cavities of the zeolite structure, there are sites where cations (positively charged ions) can be accommodated. The exchange of cations at these sites is a key feature of zeolites and is essential for their ion-exchange capacity.
- Aluminum Substitution: Zeolite structures can have aluminum atoms partially substituted for silicon atoms in the tetrahedral units. This substitution introduces a net negative charge into the framework, making zeolites overall negatively charged and allowing them to attract and exchange cations.
- Crystallinity: Zeolites exhibit high crystallinity, which means that their atoms are arranged in a well-ordered, repeating pattern. This crystalline structure is responsible for the zeolites’ stability and consistency in their properties.
- Molecular Sieve Effect: The uniform size and shape of the channels and cavities in zeolite structures result in a molecular sieve effect. This means that zeolites selectively allow molecules of a specific size and shape to enter their pores while excluding larger or smaller molecules. This property is vital for various separation and purification processes.
Zeolites can have different framework types, each characterized by a specific arrangement of tetrahedral units and channel systems. Some common zeolite framework types include zeolite A, zeolite X, and zeolite Y, among others.
Overall, the unique and highly organized crystal structure of zeolites is what makes them such valuable materials in various industrial applications, including catalysis, gas separation, and ion exchange. Their ability to selectively interact with molecules and ions within their porous framework is at the core of their utility in numerous fields.
Physical and Chemical Properties

Zeolites exhibit a wide range of physical and chemical properties that make them unique and valuable in various applications. Here are some of the key physical and chemical properties of zeolites:
Physical Properties:
- Porous Structure: Zeolites have a highly porous structure with regularly sized channels and cavities. These pores give them a high surface area, which is crucial for adsorption and ion exchange.
- High Surface Area: Zeolites typically have a high specific surface area due to their porous nature, allowing for a large contact area between the zeolite and the substances they interact with.
- Molecular Sieve Effect: Zeolites act as molecular sieves, selectively allowing molecules of specific sizes and shapes to enter their pores while excluding larger or smaller molecules. This property is essential for separation and purification processes.
- Cation Exchange Capacity: Zeolites have a strong ability to exchange cations (positively charged ions) in their structure with other cations from the surrounding environment. This property is useful for ion-exchange applications.
- Crystallinity: Zeolites exhibit a highly crystalline structure, ensuring the regular and ordered arrangement of their atoms. This crystalline nature contributes to their stability and consistency in properties.
- Thermal Stability: Zeolites can withstand high temperatures without losing their structural integrity, which is crucial in various high-temperature industrial processes.
- Hydrophilicity: Zeolites are typically hydrophilic, meaning they have an affinity for water. This property is utilized in applications related to water adsorption and purification.
Chemical Properties:
- Silicon-Aluminum Framework: The basic framework of zeolites consists of silicon (Si) and aluminum (Al) atoms connected by oxygen (O) atoms. This framework structure can vary based on the specific zeolite type.
- Acidic Nature: Zeolites can have acidic sites on their surfaces, which makes them valuable as solid acid catalysts in various chemical reactions, including hydrocarbon cracking and isomerization.
- Ion-Exchange Capacity: Zeolites can exchange cations within their structure with other cations in solution. This property is utilized in applications such as water softening and the removal of heavy metal ions from wastewater.
- Adsorption Capacity: Zeolites can adsorb various substances, including gases, liquids, and organic molecules. Their ability to selectively trap and release specific molecules is important in separation and purification processes.
- Aluminum Substitution: Aluminum atoms can partially replace silicon atoms in the tetrahedral units of zeolites, introducing a net negative charge into the framework. This charge is responsible for their cation exchange capacity.
- Dehydration Properties: Zeolites are often used as desiccants to remove moisture from gases and liquids due to their ability to adsorb water molecules effectively.
- Catalytic Activity: Some zeolites exhibit catalytic activity, particularly those with acidic sites on their surfaces. They are employed as catalysts in various chemical and petrochemical processes.
In summary, zeolites possess a unique combination of physical and chemical properties that make them versatile materials in applications ranging from water purification and wastewater treatment to catalysis, gas separation, and ion exchange. Their porous, crystalline structure and ion-exchange capabilities are at the core of their utility in a wide range of industries.
Natural Occurrence of Zeolite Minerals

Zeolite minerals are found in various geological settings around the world. They occur naturally as crystalline deposits in volcanic rocks, sedimentary rocks, and certain mineral deposits. The geological processes that lead to the formation of zeolites include volcanic activity, hydrothermal alteration, and sedimentary diagenesis. Here are some key points regarding the natural occurrence of zeolite minerals:
Geographical Distribution:
Zeolite deposits are distributed globally, and they can be found on every continent. Some regions are particularly known for their rich zeolite occurrences. Geographical locations with notable zeolite deposits include:
- Western United States: The American West, especially states like California, Oregon, and Nevada, contains significant zeolite deposits. The huge variety of zeolites found here includes clinoptilolite, mordenite, and natrolite.
- Italy: Italy is home to zeolite-rich areas, such as the volcanically active regions around Mount Vesuvius and the Aeolian Islands. Some Italian zeolites include phillipsite, chabazite, and harmotome.
- India: India has several zeolite occurrences, with notable deposits in the Deccan Plateau. These deposits contain heulandite, stilbite, and others.
- New Zealand: Zeolites can be found in parts of New Zealand, particularly the North Island. Here, minerals like stilbite and scolecite are common.
- Turkey: Turkey has zeolite deposits, including clinoptilolite and mordenite, found in various regions.
- Russia: Zeolite occurrences can be found in Russia, particularly in the eastern part of the country. Natrolite, scolecite, and mesolite are some of the zeolites discovered in Russian deposits.
Types of Zeolites Found in Nature:

Various types of zeolite minerals can be found in nature, each with its distinct crystal structure and properties. Some of the common zeolites encountered in natural deposits include:
- Clinoptilolite: Clinoptilolite is one of the most widespread and well-known natural zeolites. It is characterized by its high cation-exchange capacity and is often found in volcanic rocks and ash deposits.
- Mordenite: Mordenite is another commonly occurring zeolite, especially in volcanic regions. It forms needle-like crystals and is known for its sorption properties.
- Phillipsite: Phillipsite is often found in volcanic rocks and is recognized for its cage-like structure. It is used in various industrial applications, including water purification.
- Stilbite: Stilbite is a zeolite that occurs in sedimentary rocks and is notable for its attractive crystal formations. It is used in the petrochemical industry and as a catalyst.
- Natrolite: Natrolite is typically found in association with volcanic rocks and is known for its high sodium content. It is used in water softening and as a molecular sieve.
- Heulandite: Heulandite is found in volcanic and sedimentary rocks. It is often associated with other zeolites and is used in a variety of industrial applications.
- Analcime: Analcime is found in alkaline basalts and is known for its cubic crystal structure. It has applications in the ceramics and construction industries.
- Chabazite: Chabazite is commonly found in volcanic rocks and sedimentary formations. It has applications in ion exchange and water purification.
- Laumontite: Laumontite is typically found in low-temperature hydrothermal deposits. It is used in the production of cement and as a filter aid in various industries.
- Scolecite: Scolecite occurs in cavities and fissures in basaltic rocks and is known for its long, slender crystals. It has applications in the purification of liquids and gases.
Mining and Extraction Processes:
Zeolites are typically mined from deposits where they occur naturally. The mining and extraction processes for zeolites involve the following steps:
- Prospecting and Exploration: Geological surveys and prospecting are carried out to identify potential zeolite deposits. This involves collecting samples and conducting mineralogical analyses.
- Drilling and Sampling: Once a potential deposit is identified, drill cores are collected to assess the quality and quantity of zeolite present.
- Mining: Zeolites are extracted from the deposits using conventional mining techniques such as open-pit or underground mining, depending on the location and depth of the deposit.
- Crushing and Grinding: The mined zeolite ore is then crushed and ground into a fine powder to facilitate further processing.
- Processing: The processed zeolite is subjected to various treatments, including washing, drying, and often, additional purification steps to remove impurities.
- Packaging and Distribution: The final product is packaged and distributed for various industrial applications, including water treatment, petrochemical processes, and more.
It’s important to note that the specific mining and extraction processes may vary depending on the type of zeolite and the location of the deposit. Additionally, the quality and purity of the zeolite product can be adjusted through processing steps to meet the requirements of different applications.
Synthetic Zeolites

Synthetic zeolites are artificially produced crystalline materials with structures similar to those of natural zeolites. They are manufactured using various methods and have a wide range of applications in industries such as petrochemicals, environmental remediation, and catalysis. Here are some of the methods of synthesizing synthetic zeolites and their advantages when compared to natural zeolites:
Methods of Synthesizing Zeolite Minerals:
- Hydrothermal Synthesis: Hydrothermal methods involve the crystallization of zeolites from a mixture of alumina and silica sources in an autoclave at elevated temperatures and pressures. Various parameters, such as temperature, pressure, and reaction time, can be controlled to tailor the properties of the synthetic zeolite.
- Sol-Gel Synthesis: Sol-gel synthesis involves the formation of a colloidal suspension (sol) of zeolite precursors, followed by gelation and crystallization. This method allows for precise control over the composition and structure of the resulting zeolite.
- Ionothermal Synthesis: Ionothermal synthesis uses ionic liquids as solvents for zeolite synthesis. This method can lead to the formation of zeolites that may be challenging to produce using traditional methods.
- Template-Directed Synthesis: In this approach, organic templates or structure-directing agents (SDAs) are used to guide the growth of zeolite crystals, influencing their size and shape. This method allows for the synthesis of specific zeolite structures.
Advantages of Synthetic Zeolites:
- Tailored Properties: Synthetic zeolites can be precisely designed and modified to have specific properties, including pore size, shape, and catalytic activity. This tailoring makes them highly adaptable for different industrial applications.
- Consistency and Purity: The synthetic process allows for better control over the purity and consistency of the zeolite product, reducing impurities and variations in properties.
- Higher Surface Area: Some synthetic zeolites can be engineered to have higher surface areas compared to many natural zeolites, which can lead to enhanced adsorption and catalytic performance.
- Customized Cation Exchange: The cation exchange capacity of synthetic zeolites can be tailored by selecting specific cations for exchange, making them suitable for various ion-exchange applications.
- Rapid Synthesis: The synthetic process is often faster than the geological formation of natural zeolites, enabling more efficient and timely production.
Comparison with Natural Zeolites:
- Structure and Composition: Synthetic zeolites are engineered with specific structures and compositions, while natural zeolites vary in structure and composition based on their geological origin.
- Purity: Synthetic zeolites tend to be purer and free from contaminants compared to natural zeolites, which can contain impurities from their geological surroundings.
- Tailorability: Synthetic zeolites offer greater control and tailorability of their properties, making them suitable for a wider range of applications.
- Consistency: The properties of synthetic zeolites are consistent from batch to batch, while natural zeolites can exhibit variations in quality and characteristics.
- Availability: Natural zeolites are abundant and widespread in nature, making them readily available in some regions, while synthetic zeolites require more controlled manufacturing processes.
- Cost: The cost of synthetic zeolites can be higher due to the precision and control involved in their production, while natural zeolites may be more cost-effective in regions where they are abundant.
In summary, synthetic zeolites offer advantages in terms of tailoring, consistency, and purity compared to natural zeolites. They are preferred when specific properties and performance are required for particular industrial applications. However, the choice between natural and synthetic zeolites depends on factors like availability, cost, and the desired application.
Industrial Applications

Zeolites find a wide range of industrial applications due to their unique properties, including their porous structure, cation exchange capacity, and molecular sieve effect. Their versatility makes them valuable in various industries. Here are some of the notable industrial applications of zeolites:
- Catalysis:
- Zeolites are used as solid acid catalysts in numerous chemical reactions, including hydrocarbon cracking, isomerization, and alkylation in the petrochemical and refining industries. They facilitate these reactions with high selectivity and efficiency.
- Petrochemical Industry:
- Zeolites are crucial in oil refining processes, such as fluid catalytic cracking (FCC), to convert crude oil into valuable products like gasoline, diesel, and petrochemical feedstocks. They help improve product quality and yield.
- Adsorbents and Desiccants:
- Zeolites are used as adsorbents to remove moisture and impurities from gases and liquids. They are employed in air conditioning, natural gas drying, and as desiccants for protecting sensitive products from moisture damage.
- Water Softening and Treatment:
- Zeolites, particularly sodium zeolites, are used to soften hard water by exchanging calcium and magnesium ions with sodium ions. They are also employed in water treatment for the removal of heavy metals and ammonia.
- Environmental Remediation:
- Zeolites are utilized in the removal of pollutants from water and soil. They can adsorb heavy metals, radioactive ions, and organic contaminants, making them valuable for environmental cleanup efforts.
- Gas Separation:
- Zeolites serve as adsorbents for gas separation, allowing the selective removal of specific gases, such as carbon dioxide, nitrogen, and oxygen, in various industrial processes, including air separation and gas purification.
- Detergent Additives:
- Zeolites, primarily zeolite A and zeolite X, are used as builders in laundry detergents. They enhance the detergency by sequestering calcium and magnesium ions in hard water, preventing them from interfering with the cleaning process.
- Nuclear Waste Storage:
- Zeolites can be employed as a host material for the encapsulation and storage of radioactive waste materials. They provide a stable and controlled environment for long-term storage.
- Catalytic Converters:
- Zeolites play a role in catalytic converters for automotive emissions control. They assist in converting harmful gases, such as carbon monoxide and nitrogen oxides, into less harmful substances.
- Pharmaceuticals and Drug Delivery:
- Zeolites are used in pharmaceuticals as carriers for controlled drug release and as excipients in drug formulations. They can improve drug stability and release rates.
- Agriculture:
- Zeolites are used in agriculture to improve soil quality and moisture retention. They also act as slow-release fertilizers, providing essential nutrients to plants over time.
- Hydrogen Production:
- Zeolites are employed in the production of hydrogen gas. They help purify and separate hydrogen from gas mixtures, making it an important component in hydrogen fuel production.
- Food and Beverage Industry:
- Zeolites can be used for decolorization, purification, and removal of impurities in food and beverage processing, such as sugar refining and ethanol production.
Zeolites continue to find new applications as researchers and industries explore their properties and versatility in solving various challenges. Their ability to selectively interact with molecules and ions within their porous framework makes them a valuable resource in addressing a wide range of industrial and environmental needs.
Environmental Applications

Zeolites have a wide range of environmental applications due to their exceptional adsorption, ion-exchange, and molecular sieve properties. They are used to address various environmental challenges, including pollution control, water treatment, and waste management. Here are some of the key environmental applications of zeolites:
- Wastewater Treatment:
- Zeolites are employed to remove heavy metals, ammonia, and other contaminants from industrial and municipal wastewater. They can effectively adsorb and exchange ions, making them valuable in the treatment of polluted water.
- Water Softening:
- Zeolites, particularly sodium zeolites, are used for water softening by exchanging calcium and magnesium ions in hard water with sodium ions. This process prevents the buildup of scale in water pipes and appliances.
- Groundwater Remediation:
- Zeolites are used to treat contaminated groundwater by adsorbing and immobilizing pollutants, including organic compounds, heavy metals, and radioactive ions. This is particularly important in cleaning up sites with environmental contamination.
- Air Quality Control:
- Zeolites are employed as adsorbents in air purification systems to remove volatile organic compounds (VOCs), ammonia, and other harmful gases. They help improve indoor and outdoor air quality.
- Radioactive Waste Management:
- Zeolites are used as a containment medium for the encapsulation and storage of radioactive waste materials. They provide a stable and controlled environment for long-term waste disposal.
- Soil Remediation:
- Zeolites can be applied to contaminated soils to improve their quality and reduce the mobility of heavy metals and other contaminants. This aids in soil remediation and reduces the risk of groundwater contamination.
- Agricultural Applications:
- Zeolites are used in agriculture to improve soil quality by enhancing water retention and cation exchange capacity. They can also act as slow-release fertilizers, providing essential nutrients to plants over time.
- Livestock and Poultry Farming:
- Zeolites can be added to animal feed to reduce ammonia emissions from livestock and poultry facilities. This helps control odor and improve air quality in confined animal feeding operations (CAFOs).
- Stormwater Management:
- Zeolites can be incorporated into stormwater management systems to adsorb and remove pollutants, such as heavy metals and nutrients, from runoff water before it enters natural water bodies.
- Oil Spill Cleanup:
- Zeolites have been used in oil spill cleanup efforts to adsorb and separate oil from water. Their high surface area and oil-selective properties make them effective in mitigating the environmental impact of oil spills.
- Landfill Leachate Treatment:
- Zeolites can help in the treatment of leachate from landfills, reducing the contamination of surrounding soils and groundwater by adsorbing harmful substances.
- CO2 Capture and Storage:
- Zeolites are being explored for carbon capture and storage (CCS) to reduce greenhouse gas emissions. They can adsorb carbon dioxide from industrial processes and power plant flue gases.
- Phytoremediation Enhancement:
- Zeolites can be used to improve phytoremediation, a process in which plants are used to remove contaminants from soil. Zeolites help enhance the absorption of contaminants by plant roots.
Zeolites play a crucial role in addressing environmental challenges by providing efficient and sustainable solutions for the treatment of water, air, and soil, as well as the management of hazardous waste materials. Their versatility and effectiveness in various environmental applications make them valuable tools for mitigating pollution and protecting natural resources.
Zeolites in Everyday Life

Zeolites have numerous practical applications in our everyday lives, often behind the scenes, contributing to various products and processes that we may not even be aware of. Here are some ways in which zeolites are used in everyday life:
- Detergents: Zeolites, such as zeolite A and zeolite X, are added to laundry detergents as water softeners. They help sequester calcium and magnesium ions in hard water, preventing them from interfering with the cleaning process and improving detergent performance.
- Air Fresheners: Zeolites can be found in air fresheners and odor control products. They adsorb and neutralize odors, helping to maintain pleasant indoor environments.
- Refrigeration and Air Conditioning: Zeolites are used in refrigeration and air conditioning systems to remove water vapor and maintain low humidity levels. This helps prevent the formation of ice and keeps the systems running efficiently.
- Cat Litter: Zeolites are used in cat litter products as moisture-absorbing agents. They help control odors and keep the litter dry for extended periods.
- Dehumidifiers: Zeolite-based desiccants are used in small dehumidifiers to extract moisture from the air, making them effective for preventing dampness in closets, basements, and other enclosed spaces.
- Ethylene Gas Absorption: Zeolites can be used in fruit and vegetable storage to adsorb ethylene gas, which is produced by some fruits and vegetables and can accelerate ripening. This helps prolong the freshness of produce.
- Natural Gas Purification: Zeolites are employed in the purification of natural gas. They adsorb impurities, such as water and carbon dioxide, to produce cleaner and more energy-efficient natural gas for heating and cooking.
- Cosmetics and Personal Care Products: Zeolites can be found in cosmetics and personal care products, including skin creams and powders. They contribute to product texture and absorb excess oils from the skin.
- Oral Care Products: Zeolites are used in some toothpaste formulations to help remove surface stains and improve cleaning efficacy.
- Food Processing: Zeolites can be used in food processing to separate and purify various compounds. For example, they help in the removal of contaminants from edible oils.
- Aquarium Filtration: Zeolites can be used as part of filtration systems in aquariums to remove ammonia and heavy metals, helping maintain water quality for aquatic life.
- Oil and Gas Industry: Zeolites are used in oil refining and the natural gas industry for the purification and separation of hydrocarbons.
- Automotive Emissions Control: Zeolites are a key component in catalytic converters in vehicles, helping reduce harmful emissions from automobile exhaust.
- Paints and Coatings: Zeolites can be used in some paints and coatings to control viscosity and improve texture.
- Microencapsulation: Zeolites are used for microencapsulation in various industries, including pharmaceuticals, fragrances, and food, to protect and control the release of active ingredients.
While not always in the forefront of our daily lives, zeolites play a significant role in enhancing the performance and efficiency of numerous products and processes, contributing to a more comfortable and convenient lifestyle.

In conclusion, zeolite minerals play a significant role in various industries and are crucial in addressing a wide range of global challenges. Their unique properties, such as their porous structure, cation exchange capacity, and molecular sieve effect, make them versatile materials with applications spanning from environmental remediation to petrochemical processes. Here’s a recap of the significance of zeolite minerals:
- Versatility: Zeolites are highly versatile materials with the ability to selectively adsorb, exchange ions, and act as molecular sieves. This versatility makes them valuable in addressing diverse industrial and environmental challenges.
- Environmental Remediation: Zeolites are essential for mitigating pollution and managing contaminated water, soil, and air. Their ability to adsorb heavy metals, organic contaminants, and radioactive ions contributes to environmental cleanup efforts.
- Water Treatment: Zeolites are used for water softening, as adsorbents in wastewater treatment, and in stormwater management, contributing to cleaner and safer water resources.
- Catalysis: Zeolites serve as solid acid catalysts in the petrochemical and refining industries, leading to improved fuel and chemical production.
- Air Quality Control: Zeolites help improve indoor and outdoor air quality by removing volatile organic compounds and odors.
- Waste Management: Zeolites play a role in nuclear waste storage and landfills, offering stable and controlled environments for long-term waste containment.
- Agriculture: In agriculture, zeolites enhance soil quality, improve water retention, and serve as slow-release fertilizers, contributing to more efficient and sustainable farming practices.
- Carbon Capture: Zeolites are being explored for carbon capture and storage (CCS) to reduce greenhouse gas emissions, offering a promising solution for mitigating climate change.
- Everyday Life: Zeolites are present in everyday products such as detergents, air fresheners, cat litter, and oral care items, enhancing our comfort and well-being.
Zeolites have the potential for further research and development, opening new avenues for innovation and addressing emerging challenges. As technology advances and our understanding of these materials deepens, we can expect the following:
- Tailored Zeolites: Further research can lead to the creation of zeolites with even more precise and customizable properties to suit specific applications.
- Environmental Solutions: Ongoing research can result in improved zeolite-based solutions for addressing environmental issues, including new methods for pollutant removal and waste management.
- Energy Efficiency: Zeolites may continue to play a vital role in enhancing energy efficiency, such as in natural gas purification and carbon capture technologies.
- Healthcare and Pharmaceuticals: Research into zeolite applications in drug delivery and medical treatments may lead to breakthroughs in the healthcare sector.
- Nanotechnology: Zeolites can be incorporated into nanomaterials for novel applications in various fields, including electronics and materials science.
In summary, zeolite minerals are valuable resources with vast potential for research and development. Their unique properties make them essential in addressing global challenges, from pollution control to energy efficiency, and offer exciting opportunities for innovation in a wide range of industries.
References
- Dana, J. D. (1864). Manual of Mineralogy… Wiley.
- Mindat.org. (2019): Mineral information, data and localities.. [online] Available at: https://www.mindat.org/ [Accessed. 2019].
- Wikipedia contributors. (2019, June 25). Zeolite. In Wikipedia, The Free Encyclopedia. Retrieved 02:10, June 27, 2019, from https://en.wikipedia.org/w/index.php?title=Zeolite&oldid=903388071




























