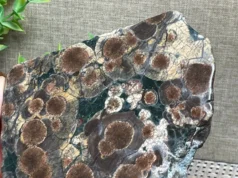
Arfvedsonite is a relatively rare mineral that belongs to the amphibole group, a subgroup of silicate minerals. Arfvedsonite is a complex inosilicate mineral with the chemical formula (Na, K)3(Fe, Mn)4Fe2Si8O22(OH)2. This formula indicates that arfvedsonite is primarily composed of sodium (Na), potassium (K), iron (Fe), manganese (Mn), silicon (Si), and oxygen (O), with hydroxyl (OH) ions in its crystal structure. It is a member of the amphibole group of minerals, which are characterized by their double chain silicate structure and a wide range of colors and physical properties.

Key characteristics of arfvedsonite include its dark green to black color, vitreous to silky luster, and prismatic or columnar crystal habit. It has a relatively high specific gravity and is known for its good cleavage in two directions. Arfvedsonite often exhibits strong pleochroism, meaning it can appear differently colored when viewed from different angles.
Historical Background and Discovery: Arfvedsonite was first described and named in 1823 by the Swedish chemist Jöns Jacob Berzelius in honor of his colleague, the Swedish chemist Johan August Arfwedson. Arfwedson had discovered the element lithium in 1817, and Berzelius named the mineral arfvedsonite as a tribute to his contributions to the field of chemistry.
Physical Properties, Chemical Composition and Crystal Structure

Physical Properties:
- Color: Arfvedsonite is typically dark green to black in color. However, it can occasionally appear brown, bluish-green, or even nearly opaque. The color variation is influenced by the presence of different elements and impurities within its crystal structure.
- Luster: Arfvedsonite exhibits a vitreous (glassy) to silky luster when polished, which enhances its overall appearance.
- Crystal Habit: Arfvedsonite commonly forms prismatic or columnar crystals with well-developed faces and a vertical striation pattern. These crystals are elongated and often occur as aggregates or bundles.
- Cleavage: It displays good cleavage in two directions at approximately 56° and 124°, which means that it can be easily split along these planes.
- Fracture: Arfvedsonite exhibits a conchoidal fracture, which results in smooth, curved surfaces when it breaks.
- Hardness: It has a relatively moderate to high hardness on the Mohs scale, typically ranging from 5.5 to 6. Arfvedsonite is hard enough to resist scratching by common household objects but can be scratched by harder minerals.
- Specific Gravity: Arfvedsonite has a specific gravity that ranges between approximately 3.3 to 3.5. This means it is denser than most common minerals.
- Transparency: It is typically translucent to opaque, with thin crystals being more translucent than thick ones.
- Pleochroism: Arfvedsonite often exhibits strong pleochroism, which means it can display different colors when viewed from different angles. This property can add to its visual appeal.
Chemical Composition: The chemical composition of arfvedsonite can be expressed by the formula (Na, K)3(Fe, Mn)4Fe2Si8O22(OH)2. This complex formula breaks down as follows:
- Sodium (Na) and potassium (K) can both be present in arfvedsonite, with sodium being more common.
- Iron (Fe) and manganese (Mn) are the primary elements responsible for its dark color.
- Silicon (Si) and oxygen (O) form the silicate tetrahedral units, which are a fundamental part of the crystal structure.
- Hydroxyl (OH) groups are also incorporated into its structure.

Crystal Structure: Arfvedsonite belongs to the amphibole group of minerals, characterized by a double chain silicate structure. Its crystal structure is monoclinic, meaning it has three unequal axes, with two of them intersecting at an oblique angle.
The crystal lattice of arfvedsonite consists of interconnected chains of silicon-oxygen tetrahedra and metal cations (primarily sodium, potassium, iron, and manganese). These chains are bonded together by additional oxygen atoms and hydroxyl groups, forming the overall structure of the mineral.
The presence of metal ions in arfvedsonite’s crystal structure contributes to its distinctive color and many of its other physical properties. The unique arrangement of atoms in its crystal lattice gives rise to its cleavage, pleochroism, and other optical characteristics.
Formation of Arfvedsonite

Arfvedsonite forms through geological processes that involve the cooling and solidification of molten rock (magma) or the transformation of pre-existing minerals under high-temperature and high-pressure conditions. Here’s more information about the formation and occurrence of arfvedsonite:
Formation: Arfvedsonite primarily forms in the following geological settings:
- Igneous Rocks: Arfvedsonite is commonly associated with igneous rocks, especially those of alkaline composition. It often crystallizes from the cooling of silica-rich, alkaline magmas that contain a variety of elements, including sodium (Na), potassium (K), iron (Fe), manganese (Mn), and silicon (Si). As the magma cools, these elements combine to form arfvedsonite crystals. The presence of iron and manganese in the magma contributes to its dark color.
- Metamorphic Rocks: In some cases, arfvedsonite can also occur in metamorphic rocks. This happens when pre-existing minerals are subjected to high-temperature and high-pressure conditions during metamorphism. Under these conditions, certain minerals can undergo chemical changes, leading to the formation of arfvedsonite.
Uses and Applications

Arfvedsonite is a relatively rare mineral, and its applications are quite limited compared to more common minerals and gemstones. However, it does have some minor uses and applications in specific contexts:
- Mineral Collecting: One of the primary uses of arfvedsonite is in mineral collecting and lapidary arts. Collectors and enthusiasts appreciate arfvedsonite for its unique crystal habit, dark color, and pleochroism, making it a desirable addition to mineral collections and jewelry pieces.
- Scientific Research: Arfvedsonite, like many other minerals, plays a role in scientific research. Geologists and mineralogists may study arfvedsonite to better understand its crystal structure, chemical composition, and the geological conditions under which it forms. This research contributes to the broader understanding of geological processes.
- Metaphysical and Healing Properties: Some individuals believe in the metaphysical or healing properties of arfvedsonite. It is sometimes associated with spiritual and holistic practices, where it is thought to possess energy-related or healing properties. These beliefs are not scientifically substantiated but are of interest to some individuals.
- Decorative Items: Arfvedsonite may occasionally be used as a decorative element in jewelry and art pieces due to its unique appearance. However, it is not a commonly used gemstone or mineral for such purposes, primarily because of its rarity and the availability of more popular gemstones.
- Lapidary Craft: In the hands of skilled lapidaries, arfvedsonite can be cut and polished to create cabochons or small gemstone-like pieces. These may be incorporated into custom jewelry designs or small-scale art projects.
It’s important to note that arfvedsonite is not widely used in industrial or technological applications due to its scarcity and the presence of other minerals with more desirable properties for such purposes. Its main value lies in its aesthetic and geological significance, making it a sought-after mineral for collectors and researchers interested in the Earth’s diverse mineralogy.
Notable Localities

Arfvedsonite is a relatively rare mineral, and its occurrence is not widespread. However, there are a few notable localities around the world where arfvedsonite has been discovered, often in association with specific geological features or occurrences. Here are some of these notable localities:
- Kola Peninsula, Russia: The Kola Peninsula in Russia is one of the most famous localities for arfvedsonite. Arfvedsonite is found in alkaline igneous rocks, including syenites and nepheline syenites, in this region. These rocks are associated with rare and unique mineral occurrences. The Kola Peninsula is known for its complex geological history and its rich diversity of mineral species, making it a prime destination for mineral collectors and researchers.
- Mont Saint-Hilaire, Canada: Mont Saint-Hilaire, located in Quebec, Canada, is another renowned locality for arfvedsonite. This site is famous for its alkaline intrusion rocks and associated minerals, including arfvedsonite. What sets Mont Saint-Hilaire apart is its extensive list of over 400 different mineral species, many of which are rare or found nowhere else in the world. The geological diversity and mineralogical richness of this location make it a significant area for both collectors and scientists.
- Sweden and Norway: Arfvedsonite has been reported in various parts of Scandinavia, including Sweden and Norway. It is found in certain alkaline rock formations in these regions, often associated with other amphibole group minerals and silicate-rich rocks. The geology of these areas has contributed to the formation of arfvedsonite and other interesting minerals.
- United States: In the United States, arfvedsonite has been reported in some localities, particularly in the western states. These occurrences are often associated with alkaline rocks and unique geological settings. While not as well-known as some of the international localities, these occurrences still contribute to the mineralogical diversity of the United States.
In these notable localities, arfvedsonite is typically found in association with specific geological features such as alkaline rock formations, syenites, and nepheline syenites. These rocks are the result of complex geological processes, and their unique chemistry and mineral assemblages create an environment conducive to the formation of arfvedsonite and other rare minerals. These localities have become important for mineral enthusiasts and researchers interested in the study and collection of rare and unusual minerals.