
Coal is a non-clastic sedimentary rock. They are the fossilized remains of plants and are in flammable black and brownish-black tones. Its main element is carbon, but it can also contain different elements such as hydrogen, sulfur and oxygen. Unlike coal minerals, it does not have a fixed chemical composition and crystal structure. Depending on the type of plant material, varying degrees of carbonization and the presence of impurities, different types of coal are formed. There are 4 recognized varieties. Lignite is the lowest grade and is the softest and least charred. Sub-bituminous coal is dark brown to black. Bituminous coal is the most abundant and is often burned for heat generation. Anthracite is the highest grade and most metamorphosed form of coal. It contains the highest percentage of low-emission carbon and would be an ideal fuel if it weren’t for comparatively less.



Coal is mainly used as a fuel. Coal has been used for thousands of years, but its real use began with the invention of steam engines after the industrial revolution. Coal provides two-fifths of electricity production worldwide and coal is used as the main fuel in iron and steel production facilities.
Name origin: The word originally took the Old English form col from the Proto-Germanic *kula(n), which is supposed to derive from the Proto-Indo-European root *g(e)u-lo- “live coal”.
Color: Black and Brownish black
Hardness: Changeable
Grain size: Fine grained
Group: Non-Clastic Sedimentary Rock
Coal Classification

As geological processes put pressure on dead biotic material over time under favorable conditions, the degree or order of metamorphic successively increases as follows:
Lignite, the lowest level of coal, the most harmful to health, is used almost exclusively as a fuel for electric power generation
Jet, a compact form of lignite, sometimes polished; Upper Paleolithic Lower-bituminous coal, whose properties range from those of lignite to bituminous coal, was primarily used as an ornamental stone as it was used as a fuel for steam-electric power generation.
Bituminous coal, a dense sedimentary rock, usually black, but sometimes dark brown, often with well-defined bands of shiny and dull material. It is primarily used as a fuel in the production of steam-electric power and in the production of coke. In the UK it is known as steam coal and has historically been used to raise steam in steam locomotives and ships.
Anthracite, the highest grade of coal, is a harder, glossy black coal used primarily for residential and commercial space heating.
Graphite is difficult to ignite and is not commonly used as a fuel; it is most commonly used in pencils or powdered for lubrication.
Channel coal (sometimes called “candle coal”) is a variety of fine-grained, high-grade coal composed primarily of liptinite with significant hydrogen content.
There are several international standards for coal. The classification of coal is generally based on the content of volatile substances. But the most important distinction is thermal coal (also known as steam coal), which is burned to generate electricity through steam; and metallurgical coal (also known as coking coal), which is burned at high temperature to make steel.
Historical significance
Coal has played an important role in human history and has been used as a source of fuel for thousands of years. In ancient times, coal was used to heat and cook food, and for warmth. During the Industrial Revolution, coal became the primary source of energy for powering steam engines and machinery, leading to significant technological advancements in transportation, manufacturing, and other industries. The use of coal also led to the development of mining as a major industry, and helped to spur economic growth in many parts of the world. However, coal use has also been associated with significant environmental impacts, including air and water pollution, and has been a major contributor to climate change. As a result, efforts are underway to transition to cleaner sources of energy and reduce dependence on coal.
Chemical composition
Coal is primarily composed of carbon, hydrogen, oxygen, nitrogen, and sulfur. The exact composition of coal varies depending on its age and origin, but generally, coal can be classified into four major types based on its carbon content: lignite, sub-bituminous, bituminous, and anthracite. Lignite is the youngest type of coal and contains the least amount of carbon, while anthracite is the oldest and has the highest carbon content. Generally, coal with higher carbon content has a higher energy content and burns more efficiently. Coal also contains varying amounts of minerals such as silica, alumina, iron, calcium, sodium, and potassium, which can affect its combustion properties and environmental impact when burned.
Physical properties

Coal has a variety of physical properties, including:
- Color: Coal can range in color from black to brown to grayish.
- Hardness: Coal can range in hardness from very soft and crumbly, like graphite, to very hard, like anthracite.
- Density: Coal has a lower density than many rocks and minerals, making it relatively lightweight.
- Porosity: Coal can be very porous, with small spaces between the coal particles.
- Conchoidal fracture: Coal often fractures in a smooth, curved pattern, known as conchoidal fracture.
- Luster: Coal has a dull to shiny luster, depending on the type of coal.
- Streak: Coal produces a black or dark brown streak when rubbed on a white, unglazed porcelain plate.
The physical properties of coal are important for its mining, processing, and use. For example, the hardness of the coal can affect the type of mining method used, while the porosity and density can affect the processing and transportation of the coal.
Mining and processing of coal

Coal is typically extracted from underground or surface mines. Underground mining methods include room and pillar, longwall, and retreat mining, while surface mining methods include strip mining, mountaintop removal, and open-pit mining.
In the room and pillar mining method, tunnels are dug into a coal seam and pillars of coal are left to support the roof. In longwall mining, a long wall of coal is mined in a single slice, while the roof over the mined-out area collapses behind the mining machine. Retreat mining involves the removal of pillars from a previously mined area.
In surface mining, the overlying rock and soil are removed to access the coal. This process can be done by strip mining, in which the overburden is removed in strips, or by mountaintop removal, in which entire mountaintops are removed to access the coal. Open-pit mining is another surface mining technique, in which a large pit is excavated to extract the coal.
Once the coal has been extracted, it is processed to remove impurities and prepare it for use. The processing may include crushing, screening, and washing to remove rock and other impurities, as well as drying to reduce the moisture content of the coal. Coal may also be treated with chemicals to remove sulfur and other impurities, a process known as coal cleaning.
Extraction techniques (surface and underground mining)
Coal mining can be divided into two broad categories: surface mining and underground mining.
Surface mining involves removing the overlying rock, soil, and vegetation to expose the coal seam. This is usually done with large machines that remove the overburden (the material above the coal seam) in layers. There are different surface mining methods, including strip mining, open-pit mining, mountaintop removal mining, and highwall mining. In strip mining, the overburden is removed in long strips, while in open-pit mining, the overburden is removed in a large pit. Mountaintop removal mining involves removing the entire top of a mountain to access the coal seam, while highwall mining is used to recover coal from an exposed vertical face or cliff.
Underground mining involves digging tunnels or shafts into the earth to reach the coal seam. There are two main types of underground mining: room and pillar mining, and longwall mining. In room and pillar mining, the coal seam is mined in a series of rooms, leaving pillars of coal to support the roof. In longwall mining, a machine called a shearer moves back and forth along the coal seam, cutting the coal and dropping it onto a conveyor belt. The roof is supported by hydraulic supports as the machine advances.
After the coal is extracted, it may be processed to remove impurities and prepared for use. The processing may involve crushing, screening, and washing to remove rocks and other materials that are mixed with the coal. The coal may also be treated with chemicals to remove sulfur and other impurities, or it may be converted to liquid or gaseous fuels.
Processing methods (cleaning, crushing, grading, etc.)
After coal is mined, it often needs to be cleaned and processed to remove impurities and prepare it for use. The exact processing methods used can vary depending on the type of coal and its intended use.
One common method of processing coal is through a process known as “washing,” which involves using water, chemicals, and mechanical equipment to separate the coal from impurities like rock, ash, and sulfur. The coal is crushed and mixed with water and chemicals to create a slurry, which is then passed through a series of screens and cyclones to separate the coal from the other materials. The separated coal is then further processed to remove any remaining impurities and graded based on size.
Other processing methods can include crushing and grinding the coal to make it suitable for burning or other uses, as well as processes to remove sulfur and other pollutants from the coal. Depending on the intended use of the coal, additional processing steps may also be required, such as carbonization to produce coke for use in the steel-making process.
Coal Composition
The composition of coal can be analyzed in two ways. The first is reported as a close analysis (moisture, volatile matter, fixed carbon and ash) or a final analysis (ash, carbon, hydrogen, nitrogen, oxygen and sulfur). A typical bituminous coal may have a final analysis on a dry, ash-free basis of 84.4% carbon, 5.4% hydrogen, 6
| ASH COMPOSİTİON, WEİGHT PERCENT | |
| SiO 2 | 20-40 |
| Al 2O 3 | 10-35 |
| Fe 2O 3 | 5-35 |
| CaO | 1-20 |
| MgO | 0.3-4 |
| TiO 2 | 0.5-2.5 |
| Na 2O & K 2O | 1-4 |
| SO 3 | 0.1-12 |
Coal Formation
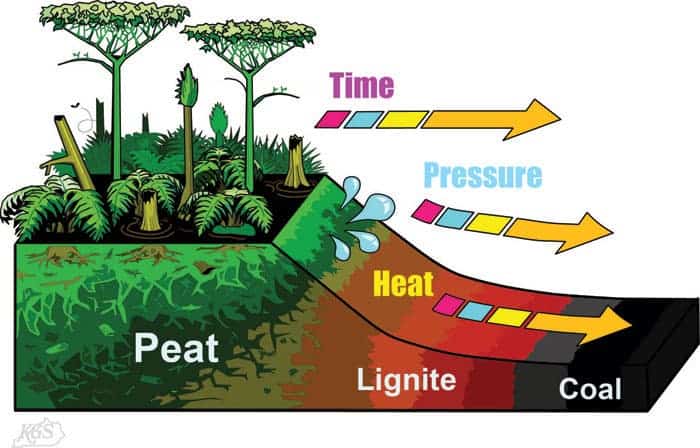
The process of turning dead vegetation into coal is called coalification. In the geological past there were low wetlands and dense forests in various regions. The dead vegetation in these areas has generally started to biodegrade and transform with mud and acidic water.
This trapped the carbon in huge peat bogs that were eventually buried deep by sediments. Then, over millions of years, the heat and pressure of the deep burial caused a loss of water, methane, and carbon dioxide and increased carbon content.
The grade of coal produced depended on the maximum pressure and temperature reached; Lignite (also called “brown coal”) and sub-bituminous coal, bituminous coal or anthracite (also called “hard coal” or “hard coal”) produced under relatively mild conditions is produced with increasing temperature and pressure.
Of the factors involved in charring, temperature is much more important than pressure or burial time. Sub-bituminous coal can form at temperatures as low as 35 to 80 °C (95 to 176 °F), while anthracite requires a temperature of at least 180 to 245 °C (356 to 473 °F).
Although coal is known from most geological periods, 90% of all coal deposits were deposited during the Carboniferous and Permian periods, which represent only 2% of Earth’s geological history.
Occurrence of Coal
Coal is a common energy and chemical source. Terrestrial plants necessary for the development of coal were not abundant until the Carboniferous period (358.9 million to 298.9 million years ago), large sedimentary basins containing rocks of Carboniferous age and younger are known on almost every continent, including Antarctica. The presence of large coal deposits in regions with currently arctic or subarctic climates (such as Alaska and Siberia) is due to climate changes and tectonic movement of crustal plates that have moved older continental masses over the Earth’s surface, sometimes through the subtropical and even tropics. regions. Some areas (like Greenland and most of northern Canada) lack coal because the rocks found there predate the Carboniferous Period, and these regions, known as continental shields, lack the abundant terrestrial plant life needed for the formation of large coal deposits.
Coal Characteristics and Properties
Many of the properties of coal vary with factors such as its composition and the presence of mineral matter. Different techniques have been developed to examine the properties of coal. These are X-ray diffraction, scanning and transmission electron microscopy, infrared spectrophotometry, mass spectroscopy, gas chromatography, thermal analysis, and electrical, thermal analysis, and electrical, optical and magnetic measurements.
Intensity
Knowing the physical properties of coal is important in the preparation and use of coal. For example, coal density ranges from about 1.1 to about 1.5 megagrams per cubic metre, or grams per cubic centimeter. Coal is slightly denser than water and significantly less dense than most rocks and mineral matter. Density differences make it possible to improve the quality of a coal by removing most of the rock matter and sulfide-rich particles through heavy liquid separation.
Porosity
Coal density is controlled in part by the presence of pores that persist throughout charring. Pore sizes and pore distribution are difficult to measure; however, pores appear to have three size ranges:
(1) macropores (diameter greater than 50 nanometers),
(2) mesopores (2 to 50 nanometers in diameter), and
(3) micropores (diameter less than 2 nanometers).
(One nanometer equals 10−9 metres.) Most of a coal’s effective surface area—about 200 square meters per gram—is found in the pores of the coal, not on the outer surface of a piece of coal. The presence of pore space is important in coke production, gasification, liquefaction and high surface area carbon production to purify water and gases. For safety reasons, coal pores may contain significant amounts of adsorbed methane, which can be released during mining operations and form explosive mixtures with air. The risk of explosion can be reduced by adequate ventilation or prior removal of coalbed methane during mining.
Reflectivity
An important property of coal is its reflectivity (or reflectivity), that is, its ability to reflect light. Reflectivity is measured by shining a monochromatic light beam (with a wavelength of 546 nanometers) onto a polished surface of vitrinite macerals in a charcoal sample and measuring the percentage of reflected light with a photometer. Vitrinite is used as its reflectivity gradually changes with increasing degree. Fusinite reflections are very high due to its coal origin and liptinites tend to disappear with increasing degrees. Although very little of the incident light is reflected (ranging from a few tenths of a percent to 12 percent), the value increases with degrees and can be used to grade most coals without measuring the percentage of volatile matter present.
Other features
Other properties such as hardness, grindability, ash fusion temperature, and free swelling index (a visual measurement of the amount of swelling that occurs when a coal sample is heated in a closed crucible) can affect coal mining and preparation. as well as the way a coal is used. Hardness and grindability determine the types of equipment used for mining, crushing and grinding, in addition to the amount of power consumed in their operations. Ash fusion temperature affects furnace design and operating conditions. The free swelling index provides preliminary information on the suitability of a coal for coke production.
Coal is an important natural resource that has played a significant role in the development of the modern world. Its economic and social importance can be seen in several areas:
- Energy production: Coal is one of the primary sources of energy used for power generation. It is burned in power plants to produce electricity, which is used to power homes, businesses, and industries.
- Steel production: Coal is also a key ingredient in the production of steel. When heated, coal releases carbon, which is used to reduce iron ore to iron. This iron is then used to produce steel, which is an essential material for construction, infrastructure, and many other applications.
- Job creation: The mining and processing of coal creates jobs and contributes to local economies in many countries. The industry employs a large number of people, including miners, engineers, geologists, and other professionals.
- Transportation: Coal is often transported long distances by rail or ship to reach its destination, which can create jobs and contribute to the economy of the areas through which it passes.
- Affordable energy: Coal is often a more affordable source of energy compared to other sources, which can help keep energy costs low for consumers and businesses.
- Chemical products: Coal is also used as a raw material in the production of a range of chemical products, including plastics, synthetic fibers, fertilizers, and other chemicals.
However, the use of coal also has significant environmental impacts, including greenhouse gas emissions and other air pollutants, as well as negative effects on water quality and land use. These impacts must be carefully considered in any evaluation of the economic and social importance of coal.
Summary of Key Points
Here are some key points about coal:
- Coal is a fossil fuel that is formed from the remains of ancient plants that lived millions of years ago.
- There are four main types of coal: lignite, sub-bituminous, bituminous, and anthracite, each with different properties and uses.
- Coal is an abundant and relatively cheap source of energy, making it an important fuel for power generation, heating, and industrial processes.
- Coal mining can have significant environmental and social impacts, including land disturbance, water pollution, and health risks for workers and nearby communities.
- Efforts are underway to develop cleaner coal technologies, such as carbon capture and storage, to reduce the environmental impact of coal use.
References
- Bonewitz, R. (2012). Rocks and minerals. 2nd ed. London: DK Publishing.
- Kopp, O. C. (2020, November 13). coal. Encyclopedia Britannica. https://www.britannica.com/science/coal-fossil-fuel
- Wikipedia contributors. (2021, October 26). Coal. In Wikipedia, The Free Encyclopedia. Retrieved 09:57, November 1, 2021, from https://en.wikipedia.org/w/index.php?title=Coal&oldid=1051971849




























