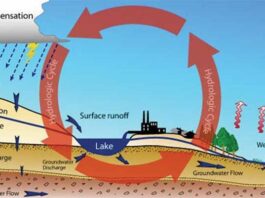
The process through which water is transferred from the surface of the Earth (land surface, free water surfaces, soil water, etc.) to the atmosphere is called evaporation.During evaporation process the latent heat of evaporation is taken from the surface of evaporation.Therefore evaporation is considered as a cooling process. Evaporation from land surface, free water surfaces, soil water, etc. are of great importance in hydrological and meterological studies,
because it affects:
- the capacity of reservoirs,
- the yield of river basins,
- the size of pumping plants,
- the consumptive use of water by plants, etc.
Transpiration defines the water loss from plants to atmosphere through the pores at the surface of their leaves.
The water returns to the atmospherein vapor form, not via a single mechanism, but through three distinct processes.
- the first process involves the fraction of water intercepted by vegetation before reaching the ground,
- the second is the transpiration of plants,
- and the third is the evaporation of gravitational water.

A portion of the precipitation falling on the vegetation covered land may be retained by plants. This portion is called interception.
This portion generally evaporates back to the atmosphere without reaching the ground surface.A very small amount of the water retained on the plants falls on the ground from the leaves. This portion is named as throughfall.
In the vegetation covered areas it is almost impossible to differentiate between evaporation and transpiration.Therefore, the two processes are lumped together and referred to as evapotranspiration.
Contents
Evaporation
Evaporation begins with the movement of molecules of water.Inside a mass of liquid water, the molecules vibrate and circulate in random fashion.This movement is related to the temperature: the higher the temperature, the more the movement is amplified.
The rate of evaporation and evapotranspiration vary depending on:
- meteorological (atmospheric) factors influencing the region,
- and on the nature of the evaporating surface.
The factors effecting the rate of evaporation (and also evapotranspiration) are:
- Solar radiation
- Relative humidity
- Air temperature
- Wind
- Atmospheric pressure
- Temperature of the liquid water
- Salinity
- Depth of water
- Aerodynamic characteristics
- Energy characteristics
Solar radiation
Solar radiation is a driving force of weather and climatic conditions, and consequently, of the hydrological cycle.Solar radiation supplies the energy necessary for the liquid water molecules to evaporate.
Solar radiation affects
- the atmosphere,
- the hydrosphere
- and the lithosphere
At the time of evaporation, thermal energy (i.e. sensible heat) is transferred into latent energy.Latent heat (energy) is the heat either absorbed or released during a phase change from ice to liquid water, or liquid water to water vapour.When water moves from liquid to gas this is a negative flux (i.e. energy is absorbed). During the opposite phase change (gas to liquid) positive heat flux occures (i.e. energy is released).
Relative humidity
For a certain temperature and air pressureit is possible to specify the maximum amount of water vapour that may be held by the parcel of air.
The saturation deficit is the difference between the saturation vapor pressure eS and the actual vapor pressure ea.
This deficit (es-ea) can also be described in relation to the concept of relative humidity Hr, Hr = (ea / es). 100
Relative humidity is the relationship between the quantity of water contained in an air mass and the maximum quantity of water the air mass can hold.
Hr = (ea / es). 100
The ability of the air to absorb more water vapor decreases as the humidity of air increases, so the rate of evaporation becomes slower.
Air temperature
Temperature is closely linked to the rate of radiation. Radiation itself is correlated directly to evaporation. It follows, then, that there is a relationship between evaporation and the temperature at the evaporating surface. The rate of evaporation is, in particular, a function of increasing temperature.Near the ground, air temperature is heavily
influenced by
- the nature of the land surface
- and the amount of sunshine.
The total amount of water vapour that may be held by a parcel of air is temperature and pressure dependent.
The temperature of air has double effect on evaporation:
- It increases saturation vapor pressure, which means increasing the saturation deficit.
- On the other hand, high temperature implies that there is energy available for evaporation.
Wind
As the liquid water vaporizes from a water body, land surface, or soil, etc.the air adjacent to these environments will become vapor saturated. For the continuation of evaporation, this saturated air should be removed. In other words atmospheric mixing has to occure.
The wind plays an essential role in the evaporation processbecause, it replaces the saturated air next to an evaporating surface with a drier layer of air. The removal of the saturated air (atmospheric mixing) is carried out by the wind.If the wind speed is zerothe parcel of air will not move away from the evaporative surface and will be saturated with water vapour.
In general, a 10% change in the wind speed causes 1-3% change in the evaporation amount when the other meteorological factors are the same.
Atmospheric pressure
Atmospheric pressure, is expressed
- in kilopascals (kPa),
- in millimeters of mercury (mm Hg)
- or in millibars (mb).
It represents the weight of a column of air per unit of area. An increase in atmospheric pressureprevents the movement of molecules out of water. The rate of evaporation increaseswhen atmospheric pressure decreases .It may be an important factor where there is an elevation difference of more than a few thousand meters.
Temperature of the liquid water
Molecular motion in the water is temperature dependent. When the temperature of the liquid water is high, molecular motion is fast. In this case the number of molecules leaving the water body will also be high, resulting an increase in evaporation.
If the temperature of the evaporating water is high, it can more readily vaporize. Thus evaporation amounts are high in tropical climates and tend to be low in polar regions. Similar contrasts are found between summer and winter evaporation quantities in mid-latitudes.
Salinity
The salinity (total dissolved solids) refers to all ions (cations and anions) dissolved in the water. The salinity of the water adversely affects evaporation. A 1% increase in salt concentration causes a 1% decrease in evaporation. A similar relationship exists with other substances in solution, because the dissolution of any substance brings about a decrease of vapor pressure. This drop in pressure is directly proportional to the concentration of the substance in solution.
Depth of water
The depth of a body of water plays a determining role in its capacity to store energy. The main difference between a shallow waterbody and a deeper one is that the shallow water is more sensitive to seasonal climatic variations. A shallow waterbody will be more sensitive to weather variations depending on the season.Deeper waterbodies, due to their thermal inertia, will have a very different evaporation response.
Aerodynamic characteristics
The aerodynamic characteristics of the surface such as
- roughness,
- texture of the material on the surface (fine or coarse materials),
- or size of the surface
also affect the amount of the evaporation.
Energy characteristics
The reflection coefficient (albedo) of the surface defines the energy characteristics of the surface.If this coefficient (albedo) is high, a larger portion of the incoming radiation will be reflected, and then evaporation will be lower from that surface.
REFERENCES
- Prof.Dr. FİKRET KAÇAROĞLU, Lecture Note, Muğla Sıtkı Koçman University
- Davie, T., 2008, Fundamentals of Hydrology (Second Ed.). Rutledge, 200 p.
- Musy, A., Higy, C., Hydrology. CRC Press, 316 p.
- Newson, M., 1994. Hydrology and The River Environment. Oxford Univ. Pres, UK, 221 p.
- Raghunath, H.M., 2006, Hydrology (Second Ed.). New Age Int. Publ., New Delhi, 463 p.
- Usul, N., Engineering Hydrology. METU Press, Ankara, 404 p.